How many grams of Ag 2 CO 3 are decomposed to yield 75.1 g Ag in this
Question:
How many grams of Ag2CO3 are decomposed to yield 75.1 g Ag in this reaction?
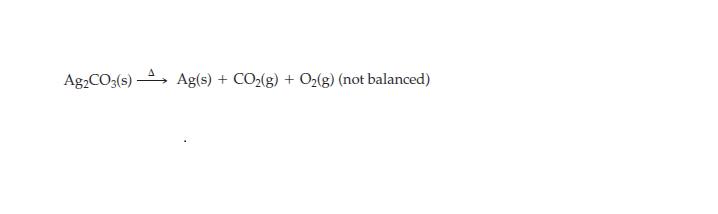
Transcribed Image Text:
Ag2CO3(s) Ag(s) + CO₂(g) + O₂(g) (not balanced)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To solve this problem we need to use stoichiometry First we need to bal...View the full answer

Answered By

John Aketch
I have a 10 years tutoring experience and I have helped thousands of students to accomplish their educational endeavors globally. What interests me most is when I see my students being succeeding in their classwork. I am confident that I will bring a great change to thins organization if granted the opportunity. Thanks
5.00+
8+ Reviews
18+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
Consider the following reaction: (a) Is this reaction exothermic or endothermic? (b) Calculate the amount of heat transferred when 3.55 g of Mg(s) reacts at constant pressure. (c) How many grams of...
-
Consider the following reaction: 2 CH3OH(g) 2 CH4(g) + O2(g).............H = + 252.8 kJ (a) Is this reaction exothermic or endothermic? (b) Calculate the amount of heat transferred when 24.0 g of...
-
The September 30, 2018, adjusted trial balance of Buzzy?s, Inc., is shown next. Requirements 1. Prepare the September closing entries for Buzzy?s, Inc. 2. Calculate the ending balance in Retained...
-
After all revenue and expense accounts have been closed at the end of the fiscal year, Income Summary has a debit of $218,380 and a credit of $375,000. At the same date, Rachel Bray, Capital has a...
-
Myers, Montgomery and Anderson-Cook (Response Surface Methodology 4th edition, Wiley, New York, 2016) discuss an experiment to determine the influence of five factors:$x_{1}$ - acid bath temperature...
-
Find the natural time period of oscillation of the pendulum shown in Fig. 13.1(a) when it oscillates between the limits \(\theta=-\pi / 2\) and \(\theta=\pi / 2\), using Eqs. (13.1) and (13.12)....
-
Rhince and Rynelf decide to merge their proprietorships into a partnership calledDawn Treader Company. The balance sheet of Rynelf Co. shows: The partners agree that the net realizable value of the...
-
Bozo Company has total costs of $100,000 when it sells 20,000 units. If total fixed costs are $40,000, what is variable cost per unit?
-
(A) Hexamethylenediamine has the molecular formula C 6 H 16 N 2 . It is one of the starting materials for the production of nylon. It can be prepared by the following reaction: C 6 H 10 O 4 (l) + NH...
-
(a) Determine the stoichiometric numbers for the following reaction, which occurs to a limited extent at 25 C: 4 KO 2 (s) + 2 CO 2 (g) 2 K 2 CO 3 (s) + 3 O 2 (g). (b) In an experiment carried out at...
-
A rigid container contains a pure mixture of liquid water and steam in equilibrium. A second species that has negligible vapor pressure is mixed into the liquid at a constant temperature. Describe...
-
How many board members did Ramalinga Raju implicate in the fraud?
-
What were the losses at HealthSouth associated with the fraud?
-
For income to be taxable, Should it be derived from legitimate sources, illegal sources, or both?
-
In Al Capones time, why would judges and law enforcement and other responsible parties ignore and condone his activities? Do persons in authority act similarly in todays society (examples)?
-
Of what crime was Al Capone convicted?
-
Shareholders of the firm Up-4-Grabs (U4G) have been offered $36.00 per share in cash for each of their U4G shares currently selling for $29.53. What is the control premium being offered in this cash...
-
Which should drive action planning more, strengths or weaknesses? That is, is it more important to build on your strengths or to reduce your weaknesses? Explain.
-
The management of an electronics manufacturing firm believes it is desirable to automate its production facility. The automated equipment would have a l0-year life with no salvage value at the end of...
-
A local symphony association offers memberships as follows: Continuing membership, per year $ 15 Patron lifetime membership 375 The patron membership has been based on the symphony association's...
-
Using capitalized cost determine which type of road surface is preferred on a particular section of highway Use 12% interest rate. A B Initial cost $700,000 $500,000 Annual maintenance Periodic...
-
Harris Company manufactures and sells a single product. A partially completed schedule of the company's total costs and costs per unit over the relevant range of 62,000 to 102,000 units is given...
-
Optimus Ltd provided you with the following statement of profit and loss and extracts from the financial position. Statement of profit or loss for the year ended 30 June 2023 Sales Cost of sales...
-
As at December 31, 2023, Bonhomme Inc. has the following shares outstanding: Common shares, 143,000 issued Preferred shares, $3.00 cumulative, 11,000 issued $1,143,000 $560,000 Dividends have not...

Study smarter with the SolutionInn App


