Identify the following types of reactions. (a) (b) (c) Cl + CHS x S.
Question:
Identify the following types of reactions.
(a)
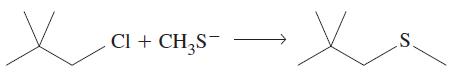
(b)
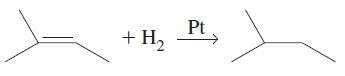
(c)
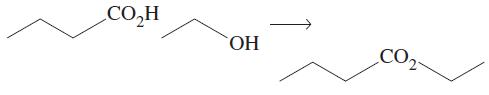
Transcribed Image Text:
Cl + CH₂S¯ x S.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
c a H3C CH3 CH3 SN CH CH3S H3C CH2 SCH3 Cl Aliphatic Nucleophilic s...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Identify the following types of reactions as precipitation, acid/base neutralization or oxidation reduction a) hcl(aq) + naoh(aq) = h2o (l) + nacl(aq) b) pb(no3) (aq) + kcl (aq) = pbbr2 (s) + kno3...
-
Identify the following types of reactions as precipitation, acid/base neutralization or oxidation reduction a. HBr (aq) + KOH (aq) H 2 O (l) + KCl(aq) b. Pb(NO 3 ) (aq) + KCl (aq) PbBr 2 (s) + KNO...
-
There are many types of information that require assurance. Individually or in groups, identify the following types of assurance services. Required: a. Identify three or more assurance services that...
-
Use any method to determine if the series converges or diverges. Give reasons for your answer. n10 10" n=1
-
Gharavi Company transferred plastic costing $3,400 and sheet metal costing $6,700 to the factory floor to be used in general maintenance projects. Because these materials will not be used in the...
-
You are given the following data: Sales $227,500,000 Invested capital $ 65,000,000 Net income .. $ 9,100,000 Compute the following: 1. Turnover of capital 2. Return on sales 3. Return on investment...
-
What do you like best about working at this company?
-
Stratton Manufacturing Company uses a standard cost accounting system. In 2011, the company produced 28,000 units. Each unit took several pounds of direct materials and 1 standard hours of direct...
-
On January 1, 2023, Blossom Ltd. acquires a building at a cost of $200,000. The building is expected to have a 20-year life and no residual value. The asset is accounted for under the revaluation...
-
(A) In each of the following reactions, determine whether the electron pair donor is acting as a BrnstedLowry base or a nucleophile. Identify the acid, electrophile, and leaving group, as...
-
Draw the structure of the transition state for the second step shown in Figure 27-14. Figure 27-14 CH3CH- OH + H Strong nucleophile CH- H H H H + a carbon is 1 -H CH3CH SN2 H + -H + HO Protonation...
-
Which category of C++ reference variables is always aliases?
-
6. The switch in the circuit shown is first thrown to point b for a long time then thrown to point a. The capacitor in the circuit shown is initially uncharged. R=40 , c = 2F, = 7 V. Find, (4-pnts)...
-
In the circuit shown above, R1 and R2 are 30 ohms and 60 ohms, respectively, and /1 = 4 amperes. The potential difference across R2 is equal to (A) 30 V (B) 60 V (C) 120 V (D) 240 V (E) a quantity...
-
QUESTION 2 The Figure Q2 shows a ball and block with masses M-10kg and M, respectively. The block and ball are connected with a cable and pulley system. The coefficient of static friction for all...
-
A 62.6 kg ice skater moving to the right with a velocity of 2.13 m/s throws a 0.179 kg snowball to the right with a velocity of 37.2 m/s relative to the ground. What is the velocity of the ice skater...
-
The position of a particle (mass = 0.15 kg) is given by: F = [(5 t - 2t) + (3 t + 5 t) ]] m, with t in s. What is the instantaneous power needed for this motion at t = 2s?
-
AstroStar, Inc., has a board of directors consisting of three members (Eckhart, Dolan, and Macero) and has approximately 500 shareholders. At a regular board meeting, the board selects Galiard as...
-
1. Below is depicted a graph G constructed by joining two opposite vertices of C12. Some authors call this a "theta graph" because it resembles the Greek letter 0. a. What is the total degree of this...
-
After all foreign and U.S. taxes, a U.S. corporation expects to receive 3 pounds of dividends per share from a British subsidiary this year. The exchange rate at the end of the year is expected to be...
-
Solitaire Machinery is a Swiss multinational manufacturing company. Currently, Solitaires financial planners are considering whether to undertake a 1-year project in the United States. The projects...
-
What is a multinational corporation? Why do firms expand into other countries?
-
Zambrano Wholesale Corporation maintains its records on a cash basis. At the end of each year the company's accountant obtains the necessary information to prepare accrual basis financial statements....
-
Premium Imports Inc. retails racing products for BMWs, Porsches, and Ferraris. The following accounts and their balances appear in the ledger of Premium Imports Inc. on November 30, the end of the...
-
In 2022, Laureen is currently single. She paid $2,760 of qualified tuition and related expenses for each of her twin daughters Sheri and Meri to attend State University as freshmen ($2,760 each, for...

Study smarter with the SolutionInn App


