Question: In the equation the missing coefficients (a) Are each 2; (b) Are each 4; (c) Can have any values as long as they are the
In the equation
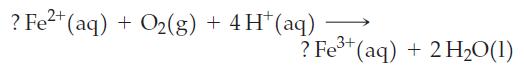
the missing coefficients
(a) Are each 2;
(b) Are each 4;
(c) Can have any values as long as they are the same;
(d) Must be determined by experiment.
? Fe+ (aq) + O(g) O(g) + 4H+ (aq) 3*(aq) + 2H,O(1) ? Fe3+
Step by Step Solution
★★★★★
3.35 Rating (158 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

The missing coefficients in the equation Fe2aq O2g Haq Fe3aq ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


