In the following diagrams, which representation demonstrates a weak electrolyte? Gas Aqueous Gas Aqueous Gas Aqueous Gas
Question:
In the following diagrams, which representation demonstrates a weak electrolyte?
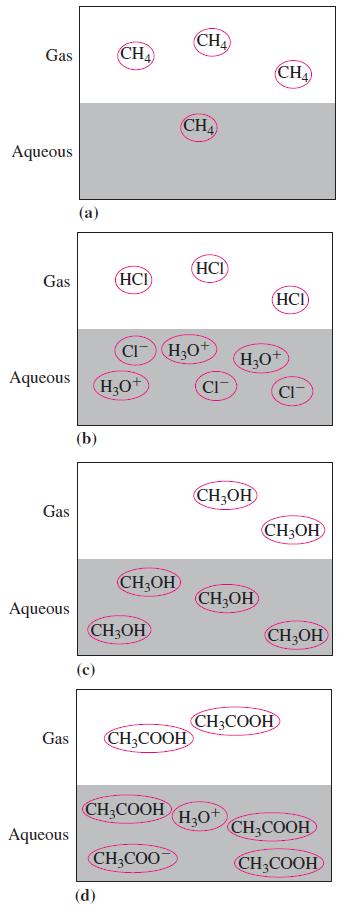
Transcribed Image Text:
Gas Aqueous Gas Aqueous Gas Aqueous Gas Aqueous (a) (b) CH4 (HCI (d) H₂O CH3OH CH₂OH CI-H₂O+ CH₂COOH CH3COOH CH₂COO CHA CHA (HCI) CI CH₂OH CH₂OH H₂O+ H3O+ CHA (HCI) CH3COOH CIT CH3OH CH₂OH CH₂COOH CH₂COOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The diagram that demonstrates a weak electrolyte is d Weak electrolytes are substances that partiall...View the full answer

Answered By

Isaiah Mutinda
As a graduate with Bs in Maths and Computer Science and having worked as a freelance full stack software developer for 3 years running I believe I have what it takes to conformable tutor and mentor a student to a professional developer also.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
In the following diagrams, which representation demonstrates a strong electrolyte? Gas Aqueous (a) Gas Aqueous Gas Aqueous (b) Aqueous (c) CHA (HCI) HO+ CHOH (d) CI) H0+ CHOH CHA Gas CH3COOH CHA...
-
Part 1 a. Ammonia, NH 3 , is a weak electrolyte. It forms ions in solution by reacting with water molecules to form the ammonium ion and hydroxide ion. Write the balanced chemical reaction for this...
-
In Problems 25 54, solve each system. Use any method you wish. 2xxy + y = 8 xy = 4
-
Wallowa Company is considering a long-term investment project called ZIP. ZIP will require an investment of $120,000. It will have a useful life of 4 years and no salvage value . Annual cash inflows...
-
The beta coefficient for Stock C is bC = 0.4 and that for Stock D is bD = 0.5. (Stock Ds beta is negative, indicating that its rate of return rises whenever returns on most other stocks fall. There...
-
Using the data in question 4, Department Xs contribution to overhead as a percentage of sales is a. 20%. c. 12%. e. 32%. b. 30%. d. 48%. Data From Question 4 A company operates three retail...
-
Paul Bergey is in charge of loading cargo ships for International Cargo Company (ICC) at the port in Newport News, Virginia. Paul is preparing a loading plan for an ICC freighter destined for Ghana....
-
How can the relationship between Online Analytical Processing (OLAP) systems and Customer Relationship Management (CRM) bolster an organization's marketing efforts?
-
An isotonic solution is described as 0.92% NaCl (mass/volume). Would this also be the required concentration for isotonic solutions of other salts, such as KCl, MgCl 2 , or MgSO 4 ? Explain.
-
NH 3 (aq)conducts electric current only weakly. The same is true for acetic acid, CH 3 COOH. When these solutions are mixed, however, the resulting solution conducts electric current very well....
-
Consider the following two-person, zero-sum game. Payoffs are the winnings for Player A. Identify the pure strategy solution. What is the value of the game? Player B b2 b3 bi a1 10 Player A
-
A nongovernment voluntary health and welfare organization (VHWO) received an unconditional pledge in 20X5 from a donor specifying that the amount pledged be used in 20X7. The donor paid the pledge in...
-
Securities donated to a voluntary health and welfare organization (VHWO) should be recorded at the a. donors recorded amount. b. fair market value at the date of the gift. c. fair market value at the...
-
A government college or university's Statement of Cash Flows would potentially report all of the following categories of cash flows except a. cash flows from operating activities. b. cash flows from...
-
GAAP require primary government total columns to be reported on which of the following basic financial statements? a. Government-Wide Statement of Net Assets if both governmental and business-type...
-
When preparing the balance sheet conversion worksheet, which of the following items must be eliminated? a. Deferred charges for debt issuance costs. b. Accrued interest payable. c. Inventories and...
-
If you were the owner of a clothes shop, how would you set about deciding what prices to charge for each garment at the end-of-season sale?
-
Show that gj concave AHUCQ Abadie For nonnegative variables, we have the following corollary.
-
Draw up an outline or flowchart tracing the capital budgeting process from the initial idea for a new investment project to the completion of the project and the start of operations. Assume the idea...
-
Compare typical compensation and incentive arrangements for (a) Top management, for example, the CEO or CFO, and (b) Plant or division managers. What are the chief differences? Can you explain them?
-
Suppose all plant and division managers were paid only a fixed salaryno other incentives or bonuses. a. Describe the agency problems that would appear in capital investment decisions. b. How would...
-
You are walking down a straight path in a park and notice there is another person walking some distance ahead of you. The distance between the two of you remains the same, so you deduce that you are...
-
The images on the right show two images. One has been taken without an aluminium fliter and the other and the second used an aluminium filter. a) Explain the purpose of an aluminium filter b)...
-
If you travel eastward from the Prime Meridian and there is a 24-minute difference between local solar noon and reference solar noon, at what longitude will you be? (1 degree longitude = 4 minutes...

Study smarter with the SolutionInn App


