Molecule (a) Below reacts faster in the S N 1 reaction than does molecule (b) Explain this
Question:
Molecule
(a) Below reacts faster in the SN1 reaction than does molecule
(b) Explain this observation. Draw chair structures for the two molecules.
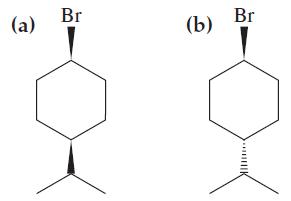
Transcribed Image Text:
(a) Br (b) Br
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Molecule (a) Below reacts faster in the S N 2 reaction than does molecule (b) Explain this observation. Draw chair structures for the most stable conformations of the two molecules. HC I (a) CH3 HC,...
-
The chemistry of derivatives of trans-decalin is of interest because this ring system is part of the structure of steroids. Make models of the brominated systems (i and ii) to help you answer the...
-
Explain why compound A reacts faster than compound B when they undergo solvolysis in aqueous acetone. CH C-Cl CH3 CH CH
-
PWX Inc. has the following information for its years ended June 30: Required: Calculate the accounts receivable turnover and average collection period for 20X3 and 20X2. Comment on the trend. What...
-
The following information is for MTC Harry Company: Beginning raw materials inventory . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . $ 30,000 Raw materials used in...
-
All sales of Tracys Jeans and Uniforms (TJU) are made on credit. Sales are billed twice monthly, on the fifth of the month for the last half of the prior months sales and on the twentieth of the...
-
1. Your instructor will divide the class into teams and assign each team the task of investigating the start-up of one of the following businesses: a. Submarine sandwich shop b. Day care service c....
-
Choosing cost drivers, activity-based costing, activity-based management. Pumpkin Bags (PB) is a designer of high quality backpacks and purses. Each design is made in small batches. Each spring, PB...
-
A 3.0-cm tall object is 55 cm in front of a diverging mirror that has a -20 cm focal length. Part A Calculate the image position. Input a positive value if the image is on the same side from the...
-
A sample of (S)-CH 3 CH 2 CH(CH 3 )Cl is hydrolyzed by water, and the resulting solution is optically inactive. (a) Write the formula of the product. (b) By which nucleophilic substitution reaction...
-
Identify the nucleophile, electrophile, and leaving group in each of the following substitution reactions. Predict whether equilibrium favors the reactants or products: (a) (b) CHCHCHCH + NaN3...
-
Kate is filling out her income tax return. Her tax for last year is $973. Her employer withheld $712 in federal taxes. a. Does Kate get a refund or does she owe the government money? b. What is the...
-
A solution is made that contains 0 . 1 4 moles of solute dissolved in 0 . 8 3 L of solution. What is the molarity of the solution? 1 . 7 5 moles of solute is dissolved in 2 2 . 4 L of solution. What...
-
A 15.67 gram sample of iron is heated in the presence of excess chlorine. A metal chloride is formed with a mass of 45.50 g. Determine the empirical formula of the metal chloride. Enter the elements...
-
C can react with CO 2 to form CO . If this reaction is applied with low processing cost, explain its impact on the chemical industry. You can explain the use of CO , the source of C and CO 2 .
-
A solution is prepared such that the initial concentration of A 2 + is 1 . 5 0 \ times 1 0 - 3 M and the initial concentration of X is 4 . 6 0 \ times 1 0 - 3 M . A 2 + and X are allowed to react as...
-
A proton has a radius of approximately 1 . 0 \ times 1 0 1 3 cm and a mass of 1 . 7 \ times 1 0 2 4 g . Part A Determine the density of a proton. For a sphere V = ( 4 3 ) \ pi r 3 .
-
Explain why someone might think this statement is true: It would be desirable to have uniform labor laws among all industrialized nations.
-
Describe the general ways that the revised Form 990, applicable for tax year 2008 and beyond, is different from previous versions.
-
Explain how to use the corporate valuation model to find the price per share of common equity.
-
Explain how it is possible for sales growth to decrease the value of a profitable company.
-
What are some actions an entrenched management might take that would harm shareholders?
-
On January 1, 2022, Quadra Limited (a company listed on the TSX stock exchange) delivered a machine to a customer that cost $50,000 to produce. The payment terms of the sale were as follows: . A down...
-
Ronald, who is in the 22% tax bracket, has come to you for advice on a potential investment. He has choice between investing in a tax exempt bond that pays 5% interest and a taxable bond that pays 7%...
-
How has U . S . GAAP developed over the years?

Study smarter with the SolutionInn App


