Predict the products of the mononitration of NO
Question:
Predict the products of the mononitration of
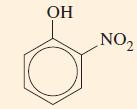
Transcribed Image Text:
ОН NO₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Analyze The groups are different and so the o pdirecting group OH guides ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Predict the mononitration products of the following aromatic compounds. (a) p-methylanisole (b) m-nitrochlorobenzene (c) p-chlorophenol (d) m-nitroanisole (e) (f) NH-C-CH CH o-methylacetanilide CHCNH...
-
Predict the mononitration products of the following compounds. (a) o-nitrotoluene (b) m-chlorotoluene (c) o-bromobenzoic acid (d) p-methoxybenzoic acid (e) m-cresol (m-methylphenol) (f)...
-
(A) Predict the major product(s) of the mononitration of benzaldehyde, C 6 H 5 CHO. (B) Predict the major product(s) of the mononitration of 1,3-dichlorobenzene.
-
Sketch the graph of the function. y = e -x/2
-
Post the following cost data to the appropriate T-accounts to trace the flow of costs from the time they are incurred until the product is completed and sold. (Assume that purchases and expenses are...
-
Several years ago, Methodist Hospital initiated its substance abuse program, which focused on counseling current and potential substance abusers. The program was funded by a grant from the state...
-
Repeat the calculations of Example 6.3, but for \(80 \mathrm{~mol} \%\) of the liquid distilled. Data From Example 6.3:- Suppose the liquid of Example 6.1 [50 mol% n-heptane (A), 50 mol% n-octane...
-
The ledger of Mai Company includes the following accounts with normal balances: D. Mai, Capital $9,000; D. Mai, Withdrawals $800; Services Revenue $13,000; Wages Expense $8,400; and Rent Expense...
-
A Rolls-Royce Phantom weighing 2.49 x 104 N is traveling in the +x direction with an initial speed of 40.2 m/s when the driver hits the brakes. The car slows to a stop in 5.60 s. If the acceleration...
-
Write equations for the substitution reaction of 1-bromobutane, a typical primary haloalkane, with the following reagents: (a) NaOH; (b) NH 3 ; (c) NaCN; (d) CH 3 CH 2 ONa.
-
In the chlorination of methane, small amounts of chloroethane will also be produced. Suggest how the formation of chloroethane might occur.
-
Define subjective performance evaluations and identify when they may be used.
-
Classify the conditions given as indicating that a reaction is at equilibrium, is not at equilibrium, or that the conditions may occur in either state. At equilibrium Not at equilibrium May or may...
-
the laboratory you are asked to make a 0 . 2 9 0 m iron ( III ) bromide solution using 3 1 5 grams of water. How many grams of iron ( III ) bromide should you add?ii
-
please solve a - d using the options found in the drop down menu a. Chair cyclohexane suffers from b. Boat cyclohexane suffers from c. Gauche butane suffers from d. Cyclopropane suffers from A Moving...
-
For the reaction A B + C, when the natural log of [A] is plotted versus the time in seconds a straight line is obtained whose slope is -0.048 s. What is the concentration of A (in M) after 13.0 s if...
-
Write Lewis structures for NF3 and PF5. On the basis of hybrid orbitals, explain the fact that NF3, PF3, and PF5 are stable molecules, but NF5 does not exist. 22. In addition to NF3, two other fluoro...
-
The FCC promulgated rules prohibiting cable television from broadcasting feature films more than 3 years old but less than 10 years old in addition to certain other programs. Home Box Office (HBO)...
-
Fred Farmer needs to prepare a balance sheet for his bank. He spent the day getting the following information. Fred needs your help to build a balance sheet and evaluate it. The information was...
-
What are the two principal reasons for holding cash? Can a firm estimate its target cash balance by summing the cash held to satisfy each of the two reasons?
-
What are the four elements of a firms credit policy? To what extent can firms set their own credit policies as opposed to accepting policies that are dictated by its competitors?
-
What are the advantages of matching the maturities of assets and liabilities? What are the disadvantages?
-
4. a) [15pts] Given the following weighted digraph D, show the matrices S,[0:5,0:5], P[0:5,0:5], k=0,...,6 as computed by Floyd's algorithm: D= 3 3411Y2 b) [5pts] Describe how the final Pmatrix P6...
-
You are tracking Chipotle Mexican Grill's stock price, attempting to figure out a good price point for entry into the market. You look at 43 random days over the course of a few months and see that...
-
Write a program that will read race times and names from a file, display the average time and then display all of the names, times and how far above or below that competitor is from the average. The...

Study smarter with the SolutionInn App


