Question: Select the correct answer and explain your reasoning. An electron having n = 3 and me = 0 (a) must have 1 ms =+; (b)
Select the correct answer and explain your reasoning.
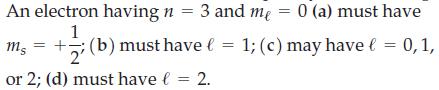
An electron having n = 3 and me = 0 (a) must have 1 ms =+; (b) must have l = 1; (c) may have = 0, 1, 2' or 2; (d) must have l = 2.
Step by Step Solution
3.46 Rating (153 Votes )
There are 3 Steps involved in it
The given information specifies that the electron has the following quantum numbers n 3 ml 3 ms ... View full answer

Get step-by-step solutions from verified subject matter experts


