State whether each of the following reactions is a substitution, an addition, an elimination, or a rearrangement
Question:
State whether each of the following reactions is a substitution, an addition, an elimination, or a rearrangement reaction. (In the equations below, H+ and Pd are catalysts.)
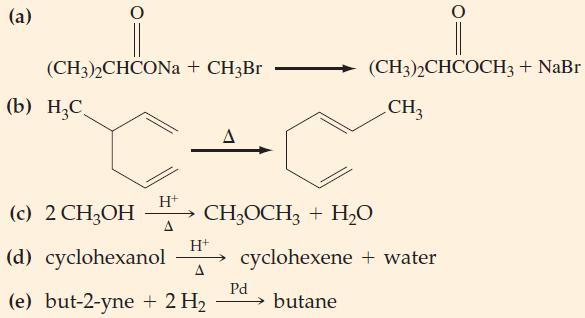
Transcribed Image Text:
(a) (CH3)2CHCONa+ CH3Br (b) H₂C H+ (c) 2 CH3OH H+ (d) cyclohexanol A (e) but-2-yne + 2 H₂ A (CH3)2CHCOCH3 + NaBr CH₂ CH3OCH3 + H₂O cyclohexene + water butane Pd
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Analyze In a substitution reaction an atom or a group of atoms is replaced with another atom or grou...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
19. Write the balanced equation for the following reactions i. Substitution reaction between methane and chlorine ii. Esterification reaction between ethanoic acid and ethanol iii. Addition reaction...
-
State whether each of the following statements is correct or incorrect concerning audit risk and its componentsinherent risk, control risk, and detection risk. a. The risk of material misstatement is...
-
State whether each of the following is or is not a principle (or a portion of a principle) underlying an audit conducted in accordance with Generally Accepted Auditing Standards. Principles Yes (Y)...
-
Give parametric equations and parameter intervals for the motion of a particle in the xy-plane. Identify the particles path by finding a Cartesian equation for it. Graph the Cartesian equation....
-
What similarities and differences exist among the costs of merchandising, manufacturing, and service firms?
-
Suppose that Mitsubishi Chemical Corporation is planning to buy new equipment to expand its production of a popular solvent. Estimated data are as follows (monetary amounts are in thousands of...
-
It would be helpful to me if I could start by asking you to basically tell me about your job. Okay?
-
Zarsons Netballs is a manufacturer of high-quality basketballs and volleyballs. Setup costs are driven by the number of batches. Equipment and maintenance costs increase with the number of...
-
Shelby Woods owns and operates an ice cream factory, the finest of its kind in all the world. Given that pints of ice cream are meant to be identical (for the same flavor), her company uses process...
-
Describe what is meant by each of the following reaction types, and illustrate with an example: (a) Nucleophilic substitution reaction; (b) Electrophilic substitution reaction; (c) Addition reaction;...
-
Are the following solvents protic or aprotic acetonitrile, CH 3 CN; ammonia, NH 3 ; trimethylamine, (CH 3 ) 3 N; formamide, HCONH 2 ; acetone, CH 3 COCH 3 .
-
Helvey brought suit against the Wabash County REMC (REMC) for breach of implied and express warranties. He alleged that REMC furnished electricity in excess of 135 volts to Helveys home, damaging his...
-
Observe the following structure and choose the correct statement. O= 1C 2C=0 3 CH3 This compound is oxidized and decarboxylated by the PDH enzyme This compound is reduced by the PDH enzyme C1 binds...
-
How easily one atom gains or loses an electron with respect to another is explained by considering the nuclear pull on valence electrons (Zeff) and how energetic/unstable valence electrons are...
-
Question 5 The term "stoichometric reaction conditions" means that _ _ _ _ _ _ _ _ _ _ _ _ _ _ . Options: equal molar amounts of reactants are used to minimize impurities. the reaction is driven to...
-
16. Calculate the daily solar energy received by a standard solar collector of dimensions 1m by 2m, in a location which receives 24MJ/m day.
-
You have a flask weighing 1 2 1 . 2 1 4 g . To this flask, you added 2 5 . 0 0 mL KCl ( aq ) solution. You weigh the flask, it is now 1 5 0 . 2 3 4 g . You then take the flask to a heating source and...
-
Explain the difference between notes, bonds, and debentures.
-
Outline some of the major problems confronting an international advertiser.
-
If a companys beta were to double, would its expected return double?
-
In the real world, is it possible to construct a portfolio of stocks that has an expected return equal to the risk-free rate?
-
An individual has $35,000 invested in a stock with a beta of 0.8 and another $40,000 invested in a stock with a beta of 1.4. If these are the only two investments in her portfolio, what is her...
-
What are the physiological adaptations of respiratory systems in diverse organisms, such as mammals, birds, and insects, and how do these adaptations optimize oxygen uptake, distribution, and...
-
Create an informal blog post about your personal experience using savings accounts to successfully save money. What are the benefits of starting early, and what kind of savings accounts can you open?...
-
Psychology is often considered a personal journey to truth. Habenicht (2000) discusses how psychology can often be a mingling of truth and error. How does this idea compete with or complement your...

Study smarter with the SolutionInn App


