The following reactions are carried out with HCl(aq) containing some tritium ( 3 1 H) as a
Question:
The following reactions are carried out with HCl(aq) containing some tritium (31H) as a tracer. Would you expect any of the tritium radioactivity to appear in the NH3(g)? In the H2O? Explain.
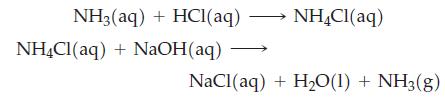
Transcribed Image Text:
NH₂(aq) + HCl(aq) →→→ NH4Cl(aq) NH4Cl (aq) + NaOH(aq) NaCl(aq) + H₂0(1) + NH3(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
In the given reactions involving HClaq containing tritium 3H its crucial to understand the fate of t...View the full answer

Answered By

Anum Naz
Lecturer and researcher with 10+ years of experience teaching courses in both undergraduate and postgraduate levels. Supervised 17 BA theses, 07 MA theses, and 1 Ph.D. dissertations. Edited and co-authored 2 monographs on contemporary trends in political thought. Published over articles in peer-reviewed journals.
4.80+
11+ Reviews
51+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
A national real-estate developer builds luxury homes in three types of locations: urban cities (city), suburbs (suburb), and rural locations that were previously farmlands (rural). The response...
-
In recent years, many companies have invested in equipment to automate processes that were once performed manually. A simple example is a drive-through car wash, where robots wash and dry the cars...
-
To what extent and how did economic factors influence the following? a. Your decision to attend college. b. Your choice of major. c. Would you expect any of these considerations to differ between men...
-
>> ((5.2^3-6.4^2+3) / (1.6^8-2))+((13.3)^1.5/(5))ans 12.2075MATLABIS THE ANSWER CORRECT? \( \gg\left(\left(5.2^{\wedge} 3-6.4^{\wedge} 2+3ight) /\left(1.6^{\wedge} 8-2ight)ight)+\left((13.3)^{\wedge}...
-
You and your partner, Kathy, own Miss Karmas Preschool, which provides preschool and day care services for about 100 children per day. Business is booming, and you are right in the middle of...
-
For the month of December 2014, Ranger Corporations records show the following information: Cash received on accounts receivable $35,000 Cash sales 30,000 Accounts receivable, December 1, 2014 80,000...
-
What are some of the nonverbal clues to deception?
-
Waldum Company purchased packaging equipment on January 5, 2012, for $135,000. The equipment was expected to have a useful life of three years, or 18,000 operating hours, and a residual value of...
-
What is a sampling distribution? Explain
-
A small quantity of NaCl containing radioactive 24 11 Na is added to an aqueous solution of NaNO 3 . The solution is cooled, and NaNO 3 is crystallized from the solution. Would you expect the NaNO 3...
-
Explain why neutron activation analysis is so useful in identifying trace elements in a sample, in contrast to ordinary methods of quantitative analysis, such as precipitation or titration.
-
Find , '2, and s2 for the random variable X that has the probability distribution f(x) = 12 for x = 2 and x = 2.
-
(a) Explain the nature and purpose of the strategic report. (150 words) (b) Discuss the benefits and potential problems of the strategic report from the viewpoint of (i) managers preparing the...
-
Draw a graph of the North American market for roses to illustrate the effects of the tariff. On the graph, identify the tariff revenue from imported roses. Wholesalers buy and sell roses in...
-
Verify the conversion factor in front of the integral in Equation 6.26. Derive the conversion factor for finding the oscillator strength from the integrated molar absorptivity according to Equation...
-
David R. and Ella M. Cole (ages 39 and 38, respectively) are husband and wife who live at 1820 Elk Avenue, Denver, CO 80202. David is a regional sales manager for Wren Industries, a national...
-
Consider the polymer: (a) Put a set of parentheses around one monomer unit, and give its molecular formula. (b) Show the line drawing for the monomer used to form this polymer. (c) Name the monomer...
-
The assignment of income doctrine states that income is taxed to the entity owning the income, regardless of who actually receives the income. That is, income taxation cannot be escaped by assigning...
-
Which property determines whether a control is available to the user during run time? a. Available b. Enabled c. Unavailable d. Disabled
-
The management of Sewell Company asks you, as the company accountant, to explain (a) The concept of reasonable assurance in internal control and (b) The importance of the human factor in internal...
-
What principle(s) of internal control is (are) involved in making daily cash counts of over-the-counter receipts?
-
Jacobs Department Stores has just installed new electronic cash registers in its stores. How do cash registers improve internal control over cash receipts?
-
A linearly elastic plane beam with a rectangular cross-section, as shown in Figure MKC7, is subjected to a bending moment of 100 kNm about its "strong" axis. NAX (mm) 200 Figure MKC7 200 200
-
Compare infinite and effective multiplication factors for three different configurations of a Cylin- drical thermal reactor with diameter D = 3.0 m and II = 4.0 m: (1) 4.5% enriched uranium moderated...
-
3-E) Using Newton's Method, select a set of coordinates, draw the complete two-sided free body diagram, identify any constraint equations, and determine the equations of motion for the adjacent...

Study smarter with the SolutionInn App


