The solubility of 1-naphthylamine, C 10 H 7 NH 2 , a substance used in the manufacture
Question:
The solubility of 1-naphthylamine, C10H7NH2, a substance used in the manufacture of dyes, is given in a handbook as 1 g per 590 g H2O. What is the approximate pH of a saturated aqueous solution of 1-naphthylamine?
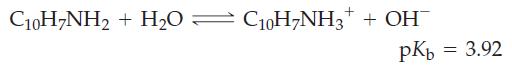
Transcribed Image Text:
C10H7NH₂ + H₂0 C₁0H7NH3+ + OH pKb = 3.92
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To determine the approximate pH of a saturated aqueous solution of 1naphthylamine we need to find th...View the full answer

Answered By

Shebla K
I am an MBA graduate having experience as an Assistant Professor at University level for two years. I always prepare well for a class as I believe that only if you become an ocean you can give a bucket of water. Being a teacher was not only my profession but also my passion.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Write a structural formula for the CH3NO isomer characterized by the structural unit indicated. None of the atoms in the final structure should have a formal charge. All the Diagrams are included in...
-
United Resources Ltd. (URL) is a mining company headquartered in Bozeman, Montana. While palladium has seen significant price increases over the past few years as it has been adopted as a critical...
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
On April 29, 2016, Auk Corporation acquires 100% of the outstanding stock of Amazon Corporation (E & P of $750,000) for $1.2 million. Amazon Corporation has assets with a fair market value of $1.4...
-
In January 2012, Teresa Leal was named treasurer of Casa de Diseno. She decided that she could best orient herself by systematically examining each area of the companys financial operations. She...
-
Figure is a system flowchart for P. Miesing and Companys purchase order event. Prepare a narrative to accompany the flowchart describing this purchase order event. Include in your narrative the...
-
How are computers and digital devices used in cybercrime?
-
A Brooks Clinic is considering investing in new heart-monitoring equipment. It has two options. Option A would have an initial lower cost but would require a significant expenditure for rebuilding...
-
Dr. Jose Rizals arrest, exile, trial, and eventual execution, primarily, was the result of his two books namely, Noli Me Tngere, and El Filibusterismo. However, his protagonist Crisostomo Ibarra,...
-
What are [H 3 O + ], [OH - ], pH, and pOH of 0.386 M CH 3 NH 2 ?
-
What are [H 3 O + ], [OH - ], pH, and pOH of 0.55 M M HClO 2 ?
-
For the given angle of rotation and coordinates of a point in the xy-coordinate system, find the coordinates of the point in the xy-coordinate system. = 45, ( -1, 3)
-
Dan Snyder wants sell his NFL franchise immediately (in USD), with no risk.He has one offer of JPY 100 Billion from a Japanese consortium, and another for GBP 700MM from a London group.Both offers...
-
Upon reviewing your finances, you realise that you have $10,000 in savings that you do not currently need to use. You decide to invest your savings in order to earn some returns and are currently...
-
(Related to Checkpoint 7.1) (Expected rate of return and risk) B. J. Gautney Enterprises is evaluating a security. One-year Treasury bills are currently paying 4.7 percent. Calculate the investment's...
-
Determine E for the Achilles Tendon Assumptions: (1) A = 2 cm2 (2) F = 5000 N (about 7 BW) (3) L = 0.5 cm = 5 mm (4) L = 2.5 cm = 25 mm
-
Estimate the ultimate tensile force needed to break a trabecular bone sample Specifically: (a) determine the force in a general formula (b) determine the force for a tibia Assumptions: (1) the bone...
-
Firms often face the problem of allocating an input in fixed supply among different products. Find the optimal crude oil allocation in the proceeding example if the profit associated with fiber were...
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
For 2010, Wiglaf Technology Company reported its most significant decline in net income in years. At the end of the year, C. S. Lewis, the president, is presented with the following condensed...
-
For 2010, Othere Technology Company initiated a sales promotion campaign that included the expenditure of an additional $20,000 for advertising. At the end of the year, George Wallace, the president,...
-
The comparative financial statements of Optical Solutions Inc. are as follows. The market price of Optical Solutions Inc. common stock was $60.00 on December 31, 2010. InstructionsDetermine the...
-
47. Lemar Ltd. has a December 31 year end. It is not a small CCPC. For 2021, its income taxes payable were $71,500, while for 2022, the amount was $93,600. For 2023, its estimated taxes payable are...
-
Prepare a statement of cash flows for 2 0 2 2 using the indirect method Additional information: 1 . Net income for 2 0 2 2 was $ 1 0 1 , 0 0 0 . 2 . Depreciation expense was $ 3 2 , 0 0 0 . 3 . Cash...
-
Brocon is proceeding with a Tier 1 Regulation A offering of common stock that will include a large number of investors who are not accredited. One of the likely investors is Carl, whose annual income...

Study smarter with the SolutionInn App


