The standard electrode potential of fluorine cannot be measured directly because F 2 reacts with water, displacing
Question:
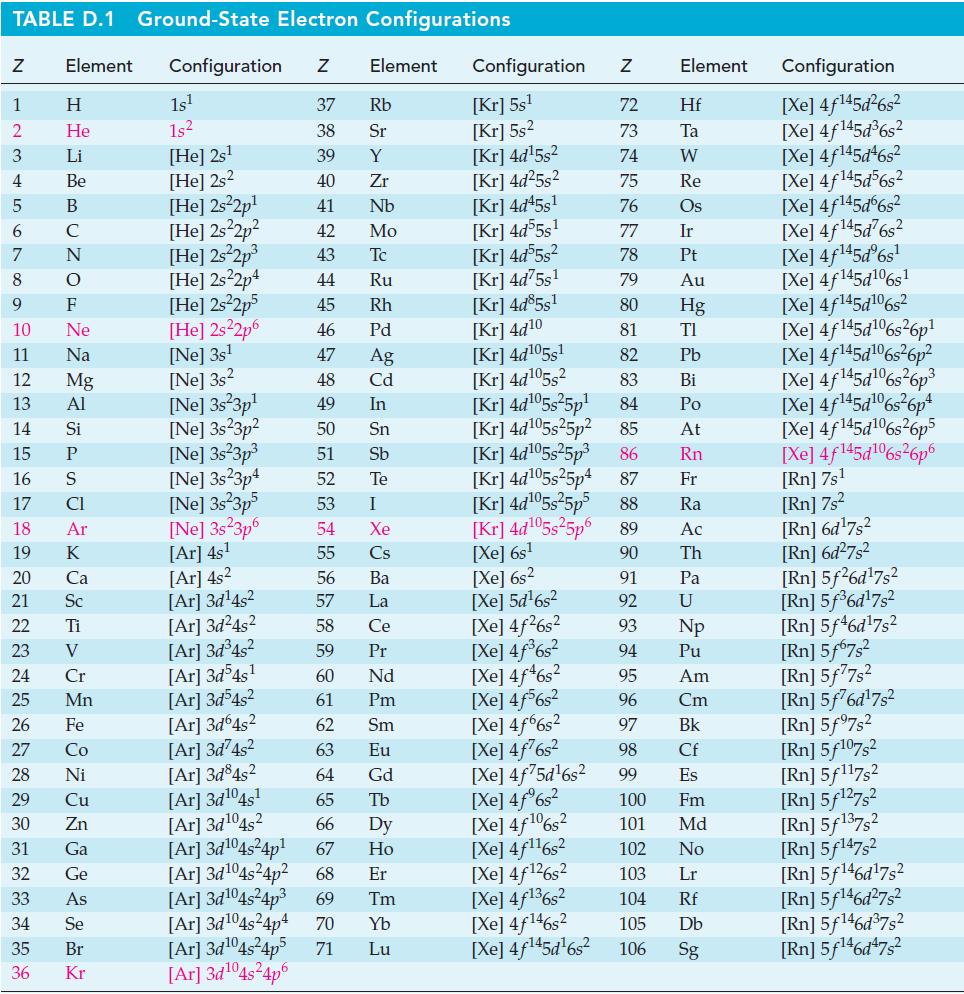
The standard electrode potential of fluorine cannot be measured directly because F2 reacts with water, displacing O2. Using thermodynamic data from Appendix D, obtain a value of E°F2/F–.
Transcribed Image Text:
TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1 2 3 4 6 7 8 9 5 B 10 11 15 16 17 18 20 25 12 13 14 Si P 27 HIG&LUZON SJE ZOSE> 0 ≤ 2 3 2 3 5 3 3 4 8 5 2 Η 29 He 30 Li 31 Be 32 33 C 19 K F Ne Na 21 Sc 22 Ti 23 V 24 Mg Al CI Ar 26 Fe Ca 28 Ni Cr Mn Co Cu Zn Ga Ge As 34 Se 35 Br 36 Kr 1s¹ 1s² [He] 2s¹ [He] 2s2 [He] 2s²2p¹ [He] 2s²2p² [He] 2s²2p³ [He] 2s22p4 [He] 2s²2p5 [He] 2s²2p6 [Ne] 3s¹ [Ne] 3s2 [Ne] 3s 3p¹ [Ne] 3s23p² [Ne] 3s²3p³ [Ne] 3s23p4 [Ne] 3s²3p5 [Ne] 3s 3p6 [Ar] 4s¹ [Ar] 4s² [Ar]3d¹4s² [Ar]3d²4s² Element 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh 46 Pd 47 Ag 48 Cd 49 In 50 Sn 51 Sb 52 Te 53 I 54 Xe 55 Cs 56 Ba 57 La 58 Ce 59 Pr 60 Nd 61 Pm 62 Sm 63 Eu 64 Gd [Ar] 3d³4s² [Ar] 3d54s¹ [Ar]3d³4s² [Ar]3d64s² [Ar]3d²4s² [Ar]3d845² [Ar]3d¹04s¹ [Ar] 3d¹04s2 65 Tb 66 67 [Ar]3d¹04s²4p¹ [Ar] 3d¹04s²4p² 68 Dy Ho Er 69 Tm [Ar]3d¹04s²4p³ [Ar] 3d¹04s²4p4 70 Yb Lu [Ar]3d¹04s²4p5 71 [Ar]3d¹04s²4p6 Configuration Z [Kr] 5s¹ [Kr] 5s² [Kr] 4d¹5s² [Kr] 4d²5s² [Kr] 4d45s¹ [kr] 4d55s¹ [Kr] 4d55s² [Kr] 4d75s¹ [Kr] 4d85s1 [Kr] 4d10 [Kr] 4d105s1 [kr] 4d¹05s² [Kr] 4d¹05s²5p¹ [Kr] 4d¹05s25p² [Kr] 4d¹05s²5p³ [kr] 4d¹05s25p4 [Xe] 6s² [Xe] 5d¹6s² [Xe] 4f²6s² [Xe] 4f³6s² [Xe] 4f46s2 [Xe] 4f6s2 [Xe] 4f6s2 [Xe] 4f²6s² [Xe] 4f75d¹6s² [Xe] 4f%s2 [Xe] 4f106s2 [Xe] 4f¹¹6s² NRNKERKR [Xe] 4f126s2 [Xe] 4f136s2 [Xe] 4f146s2 [Xe] 4f¹45d¹6s² 72 Hf 73 Ta W 74 75 Re 76 Os 77 Element 78 79 80 81 82 83 84 85 86 87 Fr [kr] 4d¹05s²5p5 88 Ra [Kr] 4d¹05s²5p6 89 Ac [Xe] 6s¹ 90 Th 91 92 93 94 95 96 97 98 99 Ir Pt Au Hg TI Pb Bi Po At Rn Pa U Np Pu Am Cm Bk Cf Es 100 Fm 101 Md 102 No 103 Lr 104 Rf 105 Db 106 Sg Configuration [Xe] 4f¹45d²6s² [Xe] 4f145d³6s² [Xe] 4f145d46s2 [Xe] 4f145d56s2 [Xe] 4f145d6s2 [Xe] 4f¹45d²6s² [Xe] 4f¹45dº6s¹ [Xe] 4f145d106s1 [Xe] 4f145d106s2 [Xe] 4f145d6s26p* [Xe] 4f145d106s36p? [Xe] 4f145d16s?6p3 [Xe] 4f145d6s®6p* [Xe] 4f145d16s26p5 [Xe] 4f145d106s26p6 [Rn] 7s¹ [Rn] 7s² [Rn] 6d¹7s² [Rn] 6d²7s² [Rn] 5f26d¹7s² [Rn] 5f³6d¹7s² [Rn] 5f46d¹7s2 [Rn] 5f67s² [Rn] 5f77s² [Rn] 5f76d¹7s² [Rn] 5f97s2 [Rn] 5f107,2 [Rn] 5f117s2 [Rn] 5f¹27s² [Rn] 5f137,2 [Rn] 5f147s2 Rn] 5f¹46d¹7s² [Rn] 5f¹46d²7s² [Rn] 5f¹46d³7s2 [Rn] 5f¹46d¹7s²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Using thermodynamic data from Appendix 4, calculate Go at 25oC for the process 2SO2(g) + O2(g) 2SO3(g) w here all gases are at 1.00 atm pressure. Also calculate Go at 25oC for this same reaction but...
-
(A) Use electrode potential data to determine G for the reaction (B) The hydrogenoxygen fuel cell is a voltaic cell with a cell reaction of 2 H 2 (g) + O 2 (g) 2 H 2 O(l). Calculate E cell for this...
-
The rest energy of many short-lived particles cannot be measured directly but must be inferred from the measured momenta and known rest energies of the decay products. Consider the (0 meson, which...
-
A pistoncylinder device contains superheated steam. During an actual adiabatic process, the entropy of the steam will (never, sometimes, always) increase.
-
The SEC requires companies to register with it when they sell stocks or bonds and also requires periodic reporting thereafter. Which of these reports, the initial registration statements or the...
-
Sow Tire, Inc., has sales of $1,450,000 and cost of goods sold of $980,000. The firm had a beginning inventory of $97,000 and an ending inventory of $82,000. What is the length of the days sales in...
-
John and Jennifer Margeson entered into a contract to sell a weight-loss franchise business called Inches-A-Weigh to Theresa Artis. The parties memorialized their agreement in an Asset Purchase...
-
Here are comparative balance sheets for Velo Company. Additional information: 1. Net income for 2014 was $93,000. 2. Cash dividends of $35,000 were declared and paid. 3. Bonds payable amounting to...
-
Structured query language (SQL) is divided into two categories: data definition language (DDL) commands and data manipulation language (DML) commands. Data manipulation language (DML) is a set of...
-
Oxygen atoms are an important constituent of the thermosphere, a layer of the atmosphere with temperatures up to 1500 K. Calculate the average translational kinetic energy of O atoms at 1500 K.
-
Zn can reduce NO 3 to NH 3 (g) in basic solution. (The following equation is not balanced.) The NH 3 can be neutralized with an excess of HCl(aq). Then, the unreacted HCl can be titrated with NaOH....
-
The dean of student activities wants to estimate the average spending on beer per week by a student. From a previous study, the standard deviation of spending was estimated to be $39. If the dean...
-
The marginal damage averted from pollution cleanup is MD = 200 5Q. The marginal cost associated with pollution cleanup is MC = 25 + 2Q. a. What is the optimal level of pollution reduction? b. Show...
-
Consider the environmental damage that results from the emissions of coal-fired power plants. These plants produce pollutants such as sulfur dioxide (SO 2 ) and nitrogen oxide (NO x ), which combine...
-
Alfie, Bill, and Coco each value police protection differently. Alfies demand for the public good is Q = 70 10P, Bills demand is Q = 70 5P, and Cocos demand is Q = 44 4P. If the marginal cost of...
-
Consider school accountability reforms like the Common Core and No Child Left Behind in the context of the results found by Deming et al. (2016), discussed in the chapter. How might Demings results...
-
Abdulkadiroglu et al. (2011) studied the effectiveness of charter schools in the Boston area using a clever research strategy. They utilized the fact that many charter schools in the area are...
-
Claudio owns a passive activity that has a basis of $28,000 and a fair market value of $38,000. The activity has suspended losses of $16,000. To reduce their estate, every year Claudio and his wife...
-
Listed below are common types of current liabilities, contingencies, and commitments: a. Accounts payable b. Bank loans and commercial paper c. Notes payable d. Dividends payable e. Sales and excise...
-
The accounting equation is: Assets = Liabilities + Stockholders Equity. Appendix A, at the end of this book, reproduces Tootsie Rolls financial statements. Replacing words in the equation with dollar...
-
Match each of the following forms of business organization with a set of characteristics: sole proprietorship (SP), partnership (P), corporation (C). (a) _____ Shared control, tax advantages,...
-
Match each of the following types of evaluation with one of the listed users of accounting information. 1. Trying to determine whether the company complied with tax laws. 2. Trying to determine...
-
Discuss how AI can affect the business by creating and sustaining competitive advantage. You can apply the strategic lenses to a company or an industry impacted by AI, and discuss how AI can be a...
-
Do you think international transportation is a major contributor to economic development and prosperity for a nation? Why? or why not? What role do governments play? Which global factors and...
-
Discuss hazardous wastes generated by businesses, industries, and institutions. The buinesses in question are dry cleaning, automotive repair, print shops, marinas and demolition companies. What kind...

Study smarter with the SolutionInn App


