Use Figure 23-8 to estimate for the reaction ZnO(s) + C(s) Zn(l) + CO(g), at 800
Question:
Use Figure 23-8 to estimate for the reaction ZnO(s) + C(s) ⇌ Zn(l) + CO(g), at 800 °C,
(a) A value of Kp;
(b) The equilibrium pressure of CO(g).
Figure 23-8
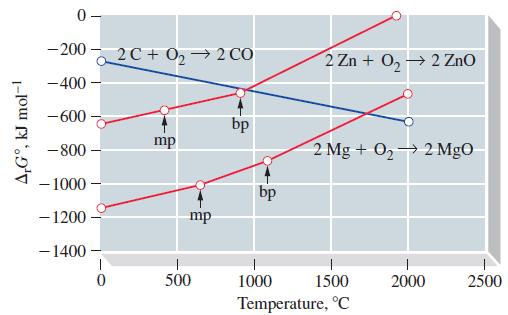
Transcribed Image Text:
A,Gº, kJ mol-1 0- -2002C+0₂ → 2 CO - 400 -600 -800- - 1000 - 1200 - 1400 0 mp mp 500 bp bp 2 Zn + 0₂ → 2 ZnO 2 Mg + O₂ 2 MgO 1000 1500 Temperature, °C 2000 2500
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a To estimate a value of Kp for the reaction ZnOs Cs Znl COg at 800 C using Figure 238 we need to fi...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Use average bond enthalpies (Table 8.4) to estimate for the atomization of benzene, C6H6: C6H6 (g) 6C (g) + 6 H (g) Compare the value to that obtained by using Hof data given in Appendix C and...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-7. Ivan sold the following securities during the year and received a Form 1099-B that...
-
I am learning intermediate accounting and how to account for leases. I have a question about the calculation of present value for the right to use of the lease. my example specifically has a...
-
Is net income under- or overstated if inventory is sold and shipped but not recorded as a sale?
-
You are the auditor for KPDZ Corporation. You gathered comparative information for inventory and accounts payable and calculated the days purchases in accounts payable. Required: Prepare a list of...
-
Tom Bonacci brought his Jeep to Brewer Service Station to investigate a strange noise the vehicle was making. The Jeep was raised up on an automobile lift so that Brewer employee Paul Gebing could...
-
Kirby Airlines is a small airline that occasionally carries overload shipments for the overnight delivery company Never-Fail, Inc. Never-Fail is a multimillion-dollar company started by Jack Never...
-
Discuss at least five of the formatting decisions you would have to make when typing a table. Compare the decisions you provided to those of two of your classmates. Do you agree or disagree with the...
-
At 400 C, r G = 25 kJ mol 1 for the reaction 2 Hg(l) + O 2 (g) 2 HgO(s). If a sample of HgO(s) is heated to 400 C, what will be the equilibrium partial pressure of O 2 (g)?
-
Use the result of the Integrative Example to determine whether a solution can be prepared with [Cu + ] equal to (a) 0.20 M; (b) 1.0 x 10 10 M. Integrative Example Although a number of slightly...
-
Would a research study on the effects of soft drink consumption on childrens cognitive development be a positive or normative statement?
-
Writing in the Wall Street Journal, economists Jeremy Siegel and Jeremy Schwartz made the following prediction: We believe that when investors awake from their depressed state, they will realize that...
-
According to an article in the New York Times, in 2012, everyone has piled into the junk bond market. The article also observed, The average yields on these bonds have dropped to 6.6 percent,...
-
An article in the Economist magazine in 2012 observed: America can now borrow from the bond market at a cheaper rate than at any time in the history of the republic. Use the loanable funds model to...
-
An article in the New York Times in 2012 observed: Older Americans and other savers are just unintended casualties of policies aimed at other economic targets, particularly the policy making it...
-
In 2012, an article in the Economist magazine recommended to investors that if economic growth and inflation remained low in the United States, the investors should buy bonds. But if inflation...
-
Astrid originally borrowed $600,000 to acquire her home. When the balance on the original mortgage is $540,000, she purchases a ski chalet by borrowing $500,000, which is secured by a mortgage on the...
-
The value of a share of common stock depends on the cash flows it is expected to provide, and those flows consist of the dividends the investor receives each year while holding the stock and the...
-
The financial statements of Zetar plc are presented in Appendix C. The companys complete annual report, including the notes to its financial statements, is available at www.zetarplc.com. Instructions...
-
Emily Frazier believes a current liability is a debt that can be expected to be paid in one year. Is Emily correct? Explain.
-
Verona Company obtains $20,000 in cash by signing a 9%, 6-month, $20,000 note payable to First Bank on July 1. Veronas fiscal year ends on September 30. What information should be reported for the...
-
On Abbot Kinney blvd, there are two coffee shops near each other. Blue bottle and intelligentsia. Blue bottle used to sell 1k cups of coffee per week. When intelligentsia lowered its prices by...
-
We use the dataset "MASchools" in the package "AER" like we did in R-Exercise 2. The dataset contains information on test performance, school characteristics and student demographic backgrounds for...
-
How involved are parents in your school? Give me three or more examples of how parents play an active role in classroom learning. Other than PTOs/PTAs, what other opportunities are available to...

Study smarter with the SolutionInn App


