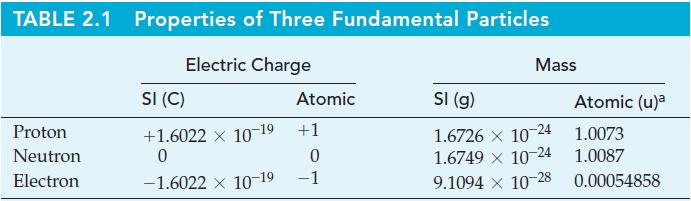
Use the electron mass from Table 2.1 and the measured mass of the nuclide 19 9 F,
Question:
Use the electron mass from Table 2.1 and the measured mass of the nuclide 199F, 18.998403 u, to determine the binding energy per nucleon (in megaelectronvolts) of this atom.
Table 2.1
Transcribed Image Text:
TABLE 2.1 Properties of Three Fundamental Particles Electric Charge Proton Neutron Electron SI (C) +1.6022 x 10-19 0 -1.6022 x 10-19 Atomic +1 0 -1 Mass SI (g) 1.6726 X 10 1.6749 × 10-24 9.1094 x 10-28 -24 Atomic (u)a 1.0073 1.0087 0.00054858
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To determine the binding energy per nucleon of the 199F atom we need to calculate the total binding ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Nucleons The term nucleon refers to the particles found in the nucleus of an atom, namely protons and neutrons. A single hydrogen atom (one proton plus one electron) has a mass of 1.007825 amu. A...
-
Verify the binding energy per nucleon given in Table for 239pu. The mass of the atom is 239.052 16u. Some Properties of Selected Nuclides Mass Binding Energy (MeV/nucleon) Spin Nuclide Stability (u)...
-
The isotope has the largest binding energy per nucleon of any isotope. Calculate this value from the atomic mass of nickel-62 (61.928345 amu) and compare it with the value given for iron-56 in Table...
-
Types of audit procedures Audit procedures are used to gatherevidence to support the auditors conclusions on the fairpresentation of a companys financial statements. Procedures can beperformed...
-
The following amounts are shown on Plymouth Companys adjusted trial balance for the year 2012: Accounts Payable . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . $ 36,000...
-
The 2013 income statement and other information for Mallard Corporation, which is about to purchase a new machine at a cost of $500 and a new computer system at a cost of $300, follows. Sales...
-
Write a code to test a Gaussian pseudorandom number generator. If you do not have a canned generator available, write a generator based on the Box-Muller algorithm in Appendix I. Apply the following...
-
First Republic Bancorp is considering the acquisition of a new data processing and management information system. The system, including computer hardware and software, will cost $1 million. Delivery...
-
1. A car has tires that have an outer diameter of 31 inches. If the wheels are turning with an angular velocity of 12 rad/s, how far in miles will the car travel in 2 hours? Enter your result rounded...
-
Using appropriate equations in the text, determine (a) The energy in joules corresponding to the destruction of 6.02 x 10 -23 g of matter; (b) The energy in megaelectronvolts that would be released...
-
What is the age of a piece of volcanic rock that has a mass ratio of argon-40 to potassium-40 of 1.9? The half-life of potassium-40 by decay is 1.26 x 10 9 years and by electron capture, 1.4 x 10 9...
-
Schaeffer Corporation reports $50 million accumulated other comprehensive income in its balance sheet as a component of shareholders' equity. In a related disclosure note reporting comprehensive...
-
(a) Draw a cubic unit cell with lattice points at (,0,0), (0,,0), (0,0,), and (,,). What is the lattice type of this unit cell and how many lattice points are contained within the unit cell? (b) Draw...
-
What is a principal-agent problem that may exist in a corporation?
-
What are the three main tasks of a financial manager?
-
Calculate the (a) foreign service premium, (b) hardship allowance, and (c) quarters allowance.
-
A presale agreement is said to be equivalent to take-out funding that may be used to repay the construction loan. What will the construction lender be concerned about if the developer plans to use...
-
Henrietta is the president and sole shareholder of Clutter Corporation. In 2008, Henrietta transferred ownership of her personal residence to the corporation. As part of the transfer, Clutter...
-
A liquid flows upward through a valve situated in a vertical pipe. Calculate the differential pressure (kPa) between points A and B. The mean velocity of the flow is 4.1 m/s. The specific gravity of...
-
Linus Hugt owns Linus Blankets. Linus asks you to explain how he should treat the following reconciling items when reconciling the companys bank account. 1. Outstanding checks 2. A deposit in transit...
-
Sue Merando is the owner of Merandos Pizza. Merandos is operated strictly on a carryout basis. Customers pick up their orders at a counter where a clerk exchanges the pizza for cash. While at the...
-
The following control procedures are used at Gonzales Company for over-the-counter cash receipts. 1. To minimize the risk of robbery, cash in excess of $100 is stored in an unlocked attach case in...
-
Balance analysis in outsourcing decisions Given the following information: In the production process of the ABX company, an intermediate product is used that is being purchased from a subcontracted...
-
On January 1, Year 3, Plaster Industries Ltd. acquired 100% of the common shares of SAX Corporation for $1,900,000. On the acquisition date, the carrying value SAX's net assets was $1,500,000. The...
-
Use the accompanying 2012 marginal tax rate table to determine the tax owed by a single man with no dependents whose gross income was $50,000. He had adjustments of $2000, and total deductions of...

Study smarter with the SolutionInn App


