Use the half-reaction method to balance the following redox equations. (a) (b) (c) C6H5NO+ Fe + H+
Question:
Use the half-reaction method to balance the following redox equations.
(a)
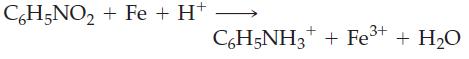
(b)
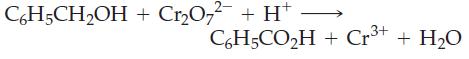
(c)
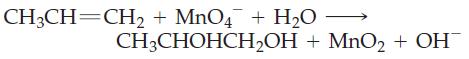
Transcribed Image Text:
C6H5NO₂+ Fe + H+ C6H5NH3 + Fe³+ + H₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
Balancing the Redox Equation Lets balance the given redox equation using the halfreaction method Equ...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Balance this redox reaction by the half reaction method by assuming an acidic solution. Cr2O72 + Fe ( Cr3+ + Fe3+
-
Balance the redox reaction in Exercise 9 by the half reaction method by assuming a basic solution. Cr2O72 + Fe ( Cr3+ + Fe3+
-
Balance the redox reaction in Exercise 11 by the half reaction method by assuming a basic solution.
-
Evaluate the geometric series or state that it diverges. 8 00 k=0 2k 75
-
The president of Radkline Corporation, Karen Pinkus, has asked you, the companys controller, to advise her on whether Radkline should develop a new inventory management system. Is the decision facing...
-
Below is a picture of a community devastated by Super Typhoon Yolanda. Analyze the picture then list down the harms it brought to the community and tell how people can cope up from it. Use a separate...
-
Hooters Restaurant in Myrtle Beach, South Carolina, used an alternative dispute resolution program, a program to resolve disputes outside the traditional court system. Employees of Hooters had to...
-
Assume that you have selected a random sample of 15 checks from a population of 800 checks. The checks you have selected are the following numbers: 664, 789, 650, 136, 365, 538, 800, 657, 110, 136,...
-
Blockett Company makes automobile sunshades and incurs the costs listed in the table below. Required: For each of the following costs, choose "Yes" to indicate if the cost is a period or category of...
-
By drawing suitable structural formulas, establish that there are 17 isomers of C 6 H 13 Cl. Refer to Example 26-1. Example 26-1 Write structural formulas for all the constitutional isomers with the...
-
Supply condensed or structural formulas for the following substances. (a) Cycloocta-1,5-diene (an intermediate in the manufacture of resins) (b) 3,7,11-trimethyl-2,6,10-dodecatriene-1-ol...
-
A simply supported beam (L = 4.5 m) must support mechanical equipment represented as a distributed load with intensity q =- 30 kN/m acting over the middle segment of the beam (see figure). Select the...
-
Suppose that one day all 330 million people in the US climb up on tables. At time t=0, we all jump off. The resulting shock, as we hit the Earth's surface, will start the entire Earth vibrating in...
-
Calculate the pH during the titration of 5 0 . 0 0 mL of 0 . 1 M acetic acid ( HOAc ) with 0 . 1 M NaOH at 2 5 C \ deg after the addition of the following volumes of reagent: ( a ) 0 mL , ( b ) 1 0 ....
-
What element is the the noble gas notation for? [Ne]3s23p O Lithium O Scandium O Neon O Aluminum
-
1.) What is at least one characteristic or property of organic compu 2.) How do we convert unsaturated compounds to saturated compounds? 3) Draw line angle formulas for ethane, propane, butane,...
-
If a constant force is applied to an area, by what factor does the pressure change if the area increases by a factor of 3? A) 9 B) 1/9 C) 3 D) No change E) 1/3
-
Describe the agents duties to the principal.
-
The following items were displayed in the statement of affairs for Lubbock Company: Fully secured liabilities ......... $90,000 Partially secured liabilities ....... 12,000 Unsecured liabilities...
-
How are secondary markets organized?
-
List some physical location markets and some computer/telephone networks.
-
Explain the differences between open outcry auctions, dealer markets, and electronic communications networks (ECNS).
-
Executive Leadership at All About Analytics realized that their talent acquisition initiatives to hire new employees from diverse cultural backgrounds are experiencing great success. The team members...
-
An investor earns a return of 6.90% per year on his investment into a portfolio of stocks in the energy sector. The inflation has has been 2.75% per year, and it is expected to stay at this level in...
-
Two corporations merge. Only one survives, Corporation B.A creditor of the non-surviving corporation, Corporation A, sues Corporation B for Corporation A's debt.Does Corporation B legally have to...

Study smarter with the SolutionInn App


