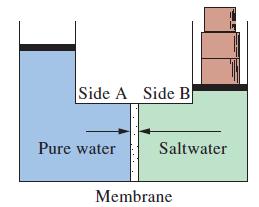
What approximate pressure is required in the reverse osmosis depicted in Figure 14-22 if the saltwater contains
Question:
What approximate pressure is required in the reverse osmosis depicted in Figure 14-22 if the saltwater contains 2.5% NaCl, by mass?
Figure 14-22
Transcribed Image Text:
Side A Side B Pure water Saltwater Membrane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The approximate pressure required in the reverse osmosis depicted in Figure 1422 if the saltwater co...View the full answer

Answered By

RAJEEV singh
I am teaching Intermediate students for last 3 years for their academics & competitive exams .
also teaching mechanical students for last 1 year .
having good grip on technical subjects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
What gauge pressure is required in the city water mains for a stream from a fire hose connected to the mains to reach a vertical height of 15.0 m? (Assume that the mains have a much larger diameter...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
Find the number density N/V for electrons such that (a) e -a = 1 and (b) e -a = 10 -6 .
-
What advantages does the profitability index provide over direct comparison of net present value when comparing two projects?
-
Duggins Veterinary Supplies can issue perpetual preferred stock at a price of $50 a share with an annual dividend of $4.50 a share. Ignoring flotation costs, what is the companys cost of preferred...
-
In general how have interest rates changed since the late 1980s according to Figure 11.1? FIGURE 11.1 Interest and Inflation Rates, 1988-2010 10p Rate (%) 8 6 10-Year Treasury Bonds 4 2 AAA Corporate...
-
Beyonce Corporation factors $175,000 of accounts receivable with Kathleen Battle Financing, Inc. on a with recourse basis. Kathleen Battle Financing will collect the receivables. The receivables...
-
A certain substance has a mass per mole of 53 g/mol. When 312 J is added as heat to a 26.0 g sample, the sample's temperature rises from 21.0C to 45.0C. What are the (a) specific heat and (b) molar...
-
Use the concentration of an isotonic saline solution, 0.92% NaCl (mass/volume), to determine the osmotic pressure of blood at body temperature, 37.0 C.
-
At 25 C a 0.50 g sample of polyisobutylene (a polymer used in synthetic rubber) in 100.0 mL of benzene solution has an osmotic pressure that supports a 5.1 mm column of solution (d = 0.88 g/mL). What...
-
In 2010, Walter Payton Company had net sales of $900,000 and cost of goods sold of $540,000. Operating expenses were $230,000, and interest expense was $11,000. Payton prepares a multiple-step income...
-
Common expenditures in a Capital Projects Fund include a. bond issue costs. b. payments to contractors. c. bond discounts. d. All of the above are common expenditures in a Capital Projects Fund. e....
-
Which of the following statements regarding the required GAAP reporting for a Capital Projects Fund is false? a. A GAAP-basis balance sheet and a Statement of Revenues, Expenditures, and Changes in...
-
Which of the following statements concerning deriving government-wide financial statements is true? a. Adjustments made to convert governmental funds to governmental activities are posted to the...
-
An Internal Service Fund established by a county is intended to operate on a breakeven basis. How might a government dispose of increases or decreases in net assets remaining at year-end?
-
Internal Service Fund net assets are sometimes increased over time as a result of charges for services being established at a level that exceeds the costs of providing the services. Why does this...
-
The adjusted trial balance for Speedy Courier as of December 31, 2013, follows. Required 1. Use the information in the adjusted trial balance to prepare (a) The income statement for the year ended...
-
A bar of length = 1 has one fixed and one free end and stiffness function c(x) = 1 - x. Find the displacement when subjected to a unit force. Pay careful attention to the boundary condition at the...
-
For small issues of common stock, the issue costs to about 10 percent of the proceeds. This means that the opportunity cost of external equity capital is about 10 percentage points higher than that...
-
There are three reasons that a common stock issue might cause a fall in price: (a) The price fall is needed to absorb the extra supply, (b) The issue causes temporary price pressure until it has been...
-
In what circumstances is a private placement preferable to a public issue? Explain.
-
3. Use the diagram to show that a price discriminating monopolist earns more profit than one charging a single price. Show further that he enjoys a greater profit the more he can discriminate (that...
-
Graph the function. (x)=x+6 Plot four points on the graph of the function: the leftmost point and three additional points.
-
Investigators did compressive strength testing on 16 different concrete formulas. Part of their data is shown in Figure 1, where eight different formulas are represented. (The only difference between...

Study smarter with the SolutionInn App


