What is the pressure (in mmHg) of the gas inside the apparatus below if P bar. =
Question:
What is the pressure (in mmHg) of the gas inside the apparatus below if Pbar. = 740 mmHg, h1 = 30 mm, and h2 = 50 mm?
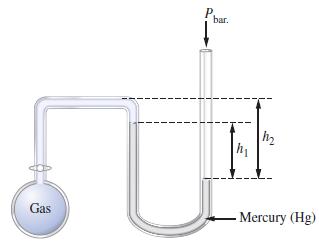
Transcribed Image Text:
Gas bar. I I I h₁ 1₂ Mercury (Hg)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To determine the pressure of the gas inside the apparatus we need to consider th...View the full answer

Answered By

Benish Ahmad
I'm a professional software engineer. I'm lectutrer at GCUF and I have 3 years of teaching experience. I'm looking forward to getting mostly computer science work including:
Programming fundamentals
Object oriented programming
Data structures
object oriented design and analysis
Database system
Computer networks
Discrete mathematics
Web application
I am expert in different computer languages such as C++, java, JavaScript, Sql, CSS, Python and C#. I'm also have excellent knowledge of essay writing and research. I have worked in other Freelancing website such as Fiverr and Upwork. Now I have finally decided to join the SolutionInn platform to continue with my explicit work of helping dear clients and students to achieve their academic dreams. I deliver plagiarism free work and exceptional projects on time. I am capable of working under high pressure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
In Figure P2.4.7, a single-reading mercury manometer is used to measure water pressure in the pipe. What is the pressure (in psi) if h1 = 6.9 in and h2 = 24.0 in.? h2 Figure P2.4.7
-
What is the pressure in kPa inside a container if it holds water that is just beginning to boil at a) 140C, b) 200C, c) 320C?
-
The atmospheric pressure at the summit of Mt. McKinley is 606 mmHg on a certain day. What is the pressure in atm and in kPa?
-
Compute the given derivatives with the help of formulas (1)(4). (a) (b) d dx (1) - x=e
-
The Tiny Treasures Company must decide whether to continue selling a line of childrens shoes manufactured on a machine that has no other purpose. The machine has a current book value of $12,000, and...
-
The following data relate to the operation of Kramer Co.s pension plan in 2016. The pension worksheet for 2015 is provided in P20-10. For 2016, Kramer will use a discount rate of 8%. Instructions (a)...
-
Mr. Prestages foot and lower leg were caught in a combine manufactured by defendant SperryNew Holland. He and his wife sued Defendant for damages arising out of the accident. Their first cause of...
-
Carlos Arruza Company exchanged equipment used in its manufacturing operations plus $3,000 in cash for similar equipment used in the operations of Tony LoBianco Company. The following information...
-
Explain briefly why countries trade If two countries, Chile and Agentina start to trade, what will determine what each country will exportor import? Explain why countries do not produce everything...
-
(A) How many moles of He(g) are in a 5.00 L storage tank filled with helium at 10.5 atm pressure at 30.0 C? (B) How many molecules of N 2 (g) remain in an ultrahigh vacuum chamber of 3.45 m 3 volume...
-
What is the volume occupied by 13.7 g Cl 2 (g) at 45 C and 98.4 kPa?
-
Find the derivative of the function. y = e 2x + e -2x
-
A small, US-based manufacturing firm is interested developing a new product to that would be produced and sold starting in 2022. Based on a preliminary market analysis, a demand forecast for the...
-
A uniform, flat, square plate has a mass of 3.0 kg and a side 2.0 m in length. The moment of inertia about an axis that passes through its center and is parallel to one edge is 1.0 kg-m. The moment...
-
Call options with an exercise price of $46 and one year to expiration are available. The market price of the underlying stock is currently $40, but this market price is expected to either decrease to...
-
Four infinite, nonconducting, thin sheets are arranged as shown. Sheet C has a uniform surface charge density of 50. The other three sheets have uniform surface charge densities of -20,-40, and 30 in...
-
Dr Anderson, a self-employed medical practitioner, incurred the following expenses during the year ended 30 June 2021: $ 1,200 4,600 800 1,200 rewiring of the surgery due to an electrical fault...
-
According to CNN, two dairy farmers challenged the legality of the funding of the Got Milk? campaigns. They argued that the Got Milk? campaigns do little to support milk from cows that are not...
-
On 1 July 2021, Croydon Ltd leased ten excavators for five years from Machines4U Ltd. The excavators are expected to have an economic life of 6 years, after which time they will have an expected...
-
(Permanent and Temporary Differences, One Rate) The accounting records of Shinault Inc. show the following data for 2010. 1. Life insurance expense on officers was $9,000. 2. Equipment was acquired...
-
(NOL without Valuation Account) Jennings Inc. reported the following pretax income (loss) and related tax rates during the years 2006' ?2012. Pretax financial income (loss) and taxable income (loss)...
-
Two Differences, Two Rates, Future Income Expected) Presented below are two independent situations related to future taxable and deductible amounts resulting from temporary differences existing at...
-
A chemical warehouse stores a collection of 40 gas cylinders consisting of 22 flammable gas cylinders and 18 inert gas cylinders. A group of 17 cylinders is randomly selected from the 40. Find the...
-
3. Mini biogasification plant was built in Selangor to convert food waste into gases under anaerobic digestion. This plant was capable to convert 500 kg of food waste into 5 kW of electricity....
-
How oversized is a carbon atom in an Fe FCC lattice in an octahedral interstitial site? The radius rFe =.124nm and rc = .077nm.

Study smarter with the SolutionInn App


