Which of the following reactions would give a better yield of CH 3 OCH(CH 3 ) 2
Question:
Which of the following reactions would give a better yield of CH3OCH(CH3)2? Explain.
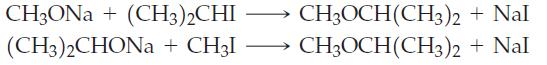
Transcribed Image Text:
CH3ONa+ (CH3)2CHI (CH3)2CHONa+ CH3I CH3OCH(CH3)2 CH3OCH(CH3)2 + Nal + Nal
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To determine which reaction would give a better yield of CH3OCHCH32 dimethyl ether lets examine the ...View the full answer

Answered By

Ishrat Khan
Previously, I have worked as an accounting scholar at acemyhomework, and have been tutoring busines students in various subjects, mostly accounting. More specifically I'm very knowledgeable in accounting subjects for college and university level. I have done master in commerce specialising in accounting and finance as well as other business subjects.
5.00+
134+ Reviews
426+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Give the major product of each of the following reactions a. b. c. d. e. f. g. h. i. CCH3 + HNO3 S NO2 CHs CH CHCH NCH CH 3 CH3 HO CH3NCH3 + PC15- NC 1. H202 2. CH3 1. HO 3. Ht CH3 + CH:CH2MgBr 2.4...
-
Give the product of each of the following reactions a. b. c. d. e. f. g. h. i. j. CHCH CH,OH -1. methanesulfonyl chloride 2. CH CO CH,CH,CH,CH,OH + PBr, Pyridine 1. p-toluenesulfonyl chloride CH:CHCH...
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
Determine which of the fundamental laws of algebra is demonstrated. 8(3 2) = 8(3) 8(2)
-
Which one of the following is not one of the four general categories of ABC activities? a. Facility support activities b. Batch-level activities c. Unit-level activities d. Product-line activities e....
-
The Ali Baba Co. is the only supplier of a particular type of Oriental carpet. The estimated demand for its carpets is Q = 112,000 500P + 5M where Q = number of carpets, P = price of carpets...
-
Based on the design, briefly discuss the data collection procedures to be used. Be sure to include the area rea of focus and targeted sample as part of these procedures. Develop a hypothetical...
-
Goodman Tire and Rubber Company has capacity to produce 170,000 tires. Goodman presently produces and sells 130,000 tires for the North American market at a price of $ 125 per tire. Goodman is...
-
Assume the company is transitioning from a traditional file environment to a database management system / relational database. - Describe and explain the challenges / issues in transitioning from a...
-
Which of the following is the strongest nucleophile for an S N 2 reaction? (a) H 2 O; (b) CH 3 CH 2 OH; (c) CH 3 CH 2 O ; (d) CH 3 CO 2 ; (e) CH 3 S .
-
(a) Which of the nucleophiles CN or Cl reacts faster with CH 3 CH 2 I in an S N 2 reaction? (b) Which of the substrates, CH 3 CH 2 CH(CH 3 )CH 2 I or CH 3 I, reacts faster with OH in an S N 2...
-
An accountant performs bookkeeping services for a trust fund. When the manager of the trust fund decided to change brokerage firms, the accountant recommended that the client shift to a particular...
-
Signal/proton type 1 Signal/proton type 2 Signal/proton type 3 A H. Chemical shift (ppm) 2.56 1.98 1.04 O B Number of protons 1 3 6 Based on the NMR spectrum, what chemical is this? C Splitting...
-
Succinic acid is a dicarboxylic acid with the formula HOOC ( CH 2 ) 2 COOH. It can be used as a standard substance for NaOH titrant standardization. To carry out the titrant standardization...
-
Mendeleev designed the first periodic table based upon the properties of elements with elements being arranged such that those with similar properties wer In fact, Mendeleev predicted the discovery...
-
Phenyl phosphate, is an artificial substrate and incubated with alkaline phosphatase in the presence of 4 mM homoarginine. The data when plotted is a Michaelis - Menton enzyme. The equation of a...
-
Ascorbic acid ( vitamin C ) content in soluble vitamin C tablets was studied using iodometric titration. For the sample solution, 1 soluble vitamin tablet with a mass of 4 . 0 0 8 8 g was taken for...
-
Defendant Atlantic Cement Company operated a large cement plant that emitted considerable amounts of dirt and smoke into the air. These emissions, combined with vibrations from the plant, caused...
-
A parking lot charges $3 for the first hour (or part of an hour) and $2 for each succeeding hour (or part), up to a daily maximum of $10. (a) Sketch a graph of the cost of parking at this lot as a...
-
As the purchaser of a new house, Carrie Underwood has signed a mortgage note to pay the Nashville National Bank and Trust Co. $8,400 every 6 months for 20 years, at the end of which time she will own...
-
Using a financial calculator, solve for the unknowns in each of the following situations. (a) On June 1, 2012, Holly Golightly purchases lakefront property from her neighbor, George Peppard, and...
-
Using a financial calculator, provide a solution to each of the following situations. (a) Lynn Anglin owes a debt of $42,000 from the purchase of her new sport utility vehicle. The debt bears annual...
-
explain the concept of chromosomal imprinting and its role in regulating gene expression in a parent-of-origin-specific manner, with potential consequences for development and disease?
-
Find the face value of the zero-coupon bond. 19-year bond at 4.3%; price $12,000 The face value will be $ (Do not round until the final answer. Then round to the nearest dollar as needed.)
-
Consider the following information for Robert Schneider's 3-stock portfolio. If the market required rate of return is currently 12 percent, and the nominal risk-free rate is 4.0 percent, what is the...

Study smarter with the SolutionInn App


