Which of the following represents a nonvolatile solute? Gas Aqueous Gas Aqueous Gas Aqueous (a) (b) Gas
Question:
Which of the following represents a nonvolatile solute?
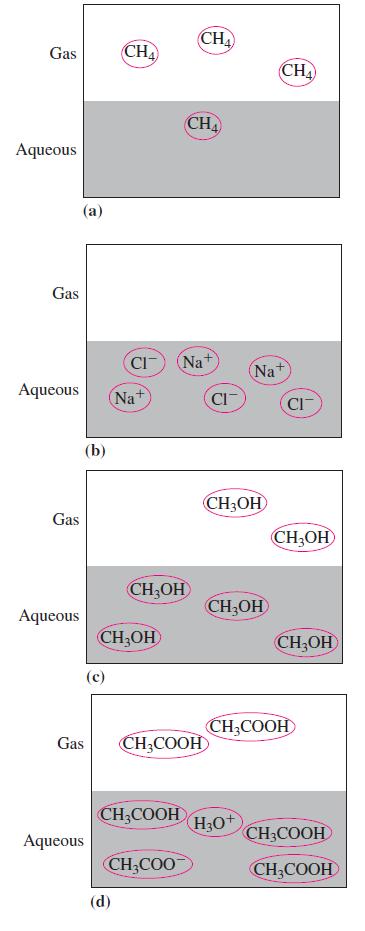
Transcribed Image Text:
Gas Aqueous Gas Aqueous Gas Aqueous (a) (b) Gas Aqueous CHA Na+ (d) CIT Na CH₂OH CH₂OH CH₂COOH CH₂ CH₂COO CHA CH₂COOH CI CH3OH Na+ CH3OH CHA H3O+ CI CH₂OH CH₂OH CH₂COOH CH3COOH CH3COOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The only nonvolatile solute is c CH3OH The other options are all volatile solutesA volatile solu...View the full answer

Answered By

Ajeet Singh
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life.
I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge.
I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields.
Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a teacher. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
4.90+
7+ Reviews
15+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
According to Table 1, the concentrations of which of the following substances varies the least with temperature? A solute is any substance that is dissolved in another substance, which is called the...
-
According to Table 1, HCl would most likely have which of the following concentrations at 70 C? A solute is any substance that is dissolved in another substance, which is called the solvent. A...
-
Which of the following represents a firms average gross receivables balance? I. Days sales in receivables x accounts receivable turnover. II. Average daily sales x average collection period. III. Net...
-
Apply the product rule for exponents, if possible. y 4 y 5 y 6
-
During the year, prepaid expenses decreased $6,500, and accrued expenses increased $2,000. Indicate how the changes in prepaid expenses and accrued expenses payable should be entered in the...
-
Recently, it was announced that two giant French retailers, Carrefour SA and Promodes SA, would merge. A headline in the Wall Street Journal blared, "French Retailers Create New Wal-Mart Rival."?...
-
Identify the most appropriate approach and then design. Provide a rationale as to why this approach and design would be most appropriate. Develop a hypothetical research scenario that would...
-
Horace Greeley Corporation was organized in 2013 and began operations at the beginning of 2014. The company is involved in interior design consulting services. The following costs were incurred prior...
-
A. Project of a building with 25 flats being evaluated for 15 years. Because of the growth for the city only 90% from the flats will be rented. This project has construction cost of 200000 and tand...
-
In a saturated solution at 25 C and 1 bar, for the following solutes, which condition will increase solubility? (a) Ar(g), decrease temperature; (b) NaCl(s), increase pressure; (c) N 2 , decrease...
-
When NH 4 Cl dissolves in a test tube of water, the test tube becomes colder. Is the magnitude of H lattice for NH 4 Cl larger or smaller than the sum of H hydration of the ions?
-
With y = (x) as in Figure 11, let Find the min and max of B on [0, 6]. A(x) = So f(t) dt and = S B(x) = f(t) dt
-
How do statutes affect the terms of contracts?
-
Ace Corporation, which is based in Texas, advertises on the Web. A court in Illinois would be most likely to exercise jurisdiction over Ace if Ace a. conducted substantial business with Illinois...
-
Apple, Inc., obtains design patents on its iPhones and iPads that cover the devices graphical user interface, shell, and screen and button design. Other patents cover the way the information is...
-
Persons suffering from mental illness are sometimes judged incapable of the state of mind required to commit a crime. (True/False)
-
Identify the element that is of prime importance in determining whether a contract has been formed.
-
Can taxes be used to relieve poverty?
-
Repeat the previous problem, but close the positions on September 20. Use the spreadsheet to find the profits for the possible stock prices on September 20. Generate a graph and use it to identify...
-
Define the market capitalization rate for a stock. Does it equal the opportunity cost of capital of investing in the stock?
-
Rework Table 4.1 under the assumption that the dividend on Fledgling Electronics is $10 next year and that it is expected to grow by 5 percent a year. The capitalization rate is 15percent. Expected...
-
In March 2001, Fly Papers stock sold for about $73. Security analysts were forecasting a long-term earnings growth rate of 8.5 percent. The company was paying dividends of $1.68 per share. a. Assume...
-
Nuperk, Inc uses a process costing system. Assembly is the the first process. They add all the raw materials needed for production at the beginning of the process. The following production...
-
If a company has total sales of forty six thousand dollars in 2023 and total sales of nine thousand in 2022, how would an analyst calculate the horizontal analysis in dollars?
-
How do emerging concepts like frugal innovation and reverse innovation redefine traditional notions of resource allocation and market penetration strategies in both developed and developing economies?

Study smarter with the SolutionInn App


