Which of the following structures are isomers? (a) CH3-CH-CH-Cl CH-CH3 (b) CH3 CH-CH-CH3 T CHCI (c) CH3-CH-CHCICH3
Question:
Which of the following structures are isomers?
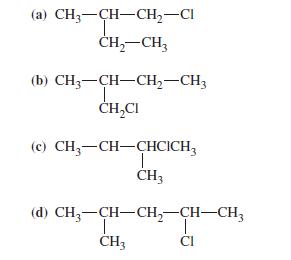
Transcribed Image Text:
(a) CH3-CH-CH₂-Cl CH₂-CH3 (b) CH3 CH-CH₂-CH3 T CH₂CI (c) CH3-CH-CHCICH3 T CH3 (d) CH3-CH-CH₂-CH-CH3 T CI CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a b c molecules are all isomers while d molecule is ...View the full answer

Answered By

Divya Munir
I hold M.Sc and M.Phil degrees in mathematics from CCS University, India and also have a MS degree in information management from Asian institute of technology, Bangkok, Thailand. I have worked at a international school in Bangkok as a IT teacher. Presently, I am working from home as a online Math/Statistics tutor. I have more than 10 years of online tutoring experience. My students have always excelled in their studies.
4.90+
0 Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
In which of the following complexes are geometric isomers possible? If isomers are possible, draw their structures and label them as cis or trans, or as fac or mer. (a) [Co(H 2 O) 4 Cl 2 ] + (b)...
-
Which of the following structures represent the same compound? Which ones represent different compounds? (a) (b) (c) (d) (e) (f) (g) Name the structures given in Problem 3-33, parts (a), (c), (e),...
-
1- A piston force F=3.5 kN is applied at point B. Find the factor- of- safety of curved beam BD. 2- For n 4, find the piston dia (d) to prevent buckling, if the length piston AB is L=600 mm. 3- Find...
-
How do you identify the potential classes in a problem domain description?
-
Calculate the present value of an $80,000, 10%, five-year bond that pays $8,000 ($80,000 10%) interest annually, if the market rate of interest is 10%. Use Exhibits 3 and 4 for computing present...
-
Compare the ISO/IEC 27001 outline with the NIST documents discussed in this chapter. Which areas, if any, are missing from the NIST documents? Identify the strengths and weaknesses of the NIST...
-
The pumper truck shown in Fig. P5.107 is to deliver \(1.5 \mathrm{ft}^{3} / \mathrm{s}\) to a maximum elevation of \(60 \mathrm{ft}\) above the hydrant. The pressure at the 4-in.-diameter outlet of...
-
(a) Estimate whether the following pairs of scores for X and Y reflect a positive relationship, a negative relationship, or no relationship. Hint: Note any tendency for pairs of X and Y scores to...
-
The proper allocation of manufacturing overhead to products produced is required by Generally Accepted Accounting Principles (GAAP) and provides a sound basis for pricing products using a full cost...
-
Give the name, condensed structural formula, and molecular mass of the molecule whose ball-and-stick model is shown. Refer to the color scheme in Figure 3-3. Figure 3-3 (a) (c) (b) (d)
-
Which of the following names is most appropriate for the molecule CH 3 (CH 2 ) 2 COOH? (a) Dimethyleneacetic acid; (b) Propanoic acid; (c) Butanoic acid; (d) Oxobutylalcohol.
-
Amanda and Raj are both students working part-time at an insurance company. Amanda can only work 5 hours a day. Her manager informs her that she needs to review 250 documents and process 250...
-
247. A tall sailing ship (m = 1.43 104 kg) is initially at rest. A steady wind blows at the 60 angle shown in the diagram and moves the ship forward a distance of 850 m. The ship's final speed is 9.0...
-
The owners of the USHL stated they can tolerate 5 - yrs of operations if their team is sustainable after that time. At the end of the 5 - yrs , which team ( s ) will cease operations? Which team ( s...
-
describe the mechanisms by which the nucleus responds to environmental stimuli or stressors, such as DNA damage, viral infections, or changes in nutrient availability?
-
Public and Private Goods In a New Year Pary, you need to decide how much money to spend on cans of beer (b, standing for the number of cans of beer) and how much to spend on fireworks (f, standing...
-
A computer call center is going to replace all of its incandescent lamps with more energy efficient fluorescent lighting fixtures. The total energy savings are estimated to be $2,104 per year, and...
-
Consider the following hypotheses: H0: p 0.38 HA: p < 0.38 Compute the p-value based on the following sample information. a. x = 22; n = 74 b. x = 110; n = 300 c. = 0.34; n = 50 d. = 0.34; n = 400
-
From 1970 to 1990, Sri Lanka's population grew by approximately 2.2 million persons every five years. The population in 1970 was 12.2 million people.What is the best formula for P, Sri Lanka's...
-
From the standpoint of the borrower, is long-term or short-term credit riskier? Explain. Would it ever make sense to borrow on a short-term basis if short-term rates were above long-term rates?
-
Firms can control their accruals within fairly wide limits. Discuss.
-
Is it true that most firms are able to obtain some free trade credit and that additional trade credit is often available, but at a cost? Explain.
-
The Excel Lessons cover all of the steps required to complete this lab. Income for this year was from Gross Sales and were $ 5 2 5 , 0 0 0 Business expenses were: Salaries $ 2 0 5 , 0 0 0 Building...
-
What is the full name of the award that would apply to a waiter in a restaurant who had several years experience and was involved in taking reservations, serving food and alcohol, placing orders,...
-
olve for the incorrect values. Please create tables and show work. During the year, TRC Corporation has the following inventory transactions. Date January 1 Transaction Beginning inventory April 7...

Study smarter with the SolutionInn App


