Question: Write condensed structural formulas for phosphoric acid and phosphorous acid. Condensed structural formulas are discussed on page 70. Structural formula: HO || H-C-C-0-H H
Write condensed structural formulas for phosphoric acid and phosphorous acid. Condensed structural formulas are discussed on page 70.
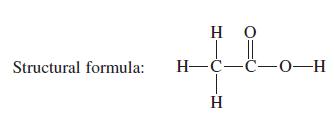
Structural formula: HO || H-C-C-0-H H
Step by Step Solution
3.34 Rating (154 Votes )
There are 3 Steps involved in it
H P OH HO P O... View full answer

Get step-by-step solutions from verified subject matter experts


