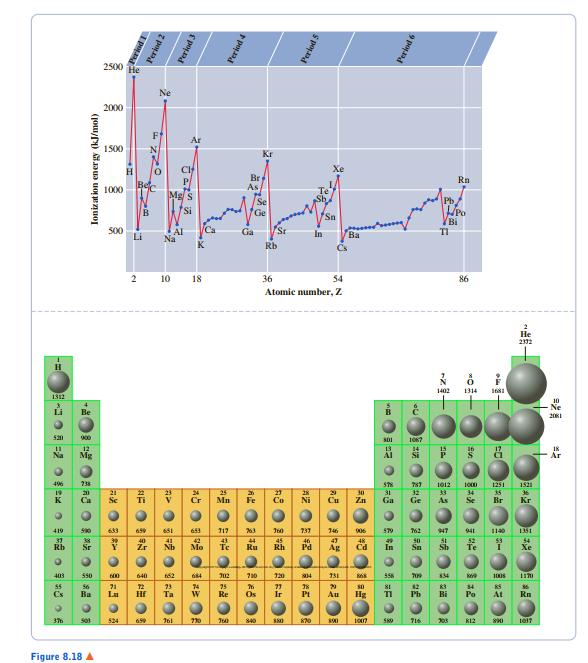
From Figure 8.18, predict the first ionization energy of francium (Z = 87). 2500 He 2000 *
Question:
From Figure 8.18, predict the first ionization energy of francium (Z = 87).
Transcribed Image Text:
2500 He 2000 * 1500 Kr Br As Rn E 1000 Bi S00 Li Ca Sr Na In Ba K Rb Cs 10 18 36 54 86 Atomic number, Z -Ne 吕 550 1008 At 524 730 J0 s40 S0 203 K12 Figure 8.18 A pona Period 2 Period 3 Ionizat ion energy (kJ/mol) Period 4 Perlod 5 つ=2の o 6 Perlod 6 AZ 中 一
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
The ionization ...View the full answer

Answered By

Wonder Dzidzormenu
As a professional accountant and a teacher, I explain account ing concepts in a more practical way that makes students more connected to the subject.
With over 10 years of teaching accounting , I offer a well constructed , easily understood and in-depth explanations to students questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
From Figure 8.18, predict the first ionization energy of ununseptium (Z = 117). 2500 He 2000 * 1500 Kr Br As Rn E 1000 Bi S00 Li Ca Sr Na In Ba K Rb Cs 10 18 36 54 86 Atomic number, Z -Ne 550 1008...
-
The first ionization energy of the chlorine atom is 1251 kJ/mol. Without looking at Figure 8.18, state which of the following values would be the more likely ionization energy for the iodine atom....
-
The first ionization energy of the oxygen molecule is the energy required for the following process: O2 (g) O2+ (g) + e- The energy needed for this process is 1175kJ/mol, very similar to the first...
-
A sample consisting of 1.00mol Ar is expanded isothermally at 20 C from 10.0dm 3 to 30.0dm 3 (i) Reversibly, (ii) Against a constant external pressure equal to the final pressure of the gas, (iii)...
-
Sport utility vehicles (SUVs), vans, and pickups are generally considered to be more prone to roll over than cars. In 1997, 24.0% of all highway fatalities involved rollovers; 15.8% of all fatalities...
-
Arnez Companys annual accounting period ends on December 31, 2019. The following information concerns the adjusting entries to be recorded as of that date. Entries can draw from the following partial...
-
Six test scores are shown below. The first 4 test scores are 15% of the final grade, and the last two test scores are 20% of the final grade. Find the weighted mean of the test scores. 80 70 84 93 89...
-
Lessee Company leases heavy equipment on January 1, 2010 under a capital lease from Lessor Company with the following lease provisions: The lease is noncancelable and has a term of 10 years. The...
-
A loan has a stated annual rate of 19.71%. If loan payments are made monthly and interest is compounded monthly, what is the effective annual rate of interest? You invest $3,679.00 at the beginning...
-
Referring to Exercise 3.39 find (a) f(y\2) for all values of y; (b) P(Y = 0 | X = 2).
-
What property of an atom does nuclear magnetic resonance depend on? What frequency range of the electromagnetic spectrum does NMR use?
-
For eka-lead, predict the electron configuration, whether the element is a metal or nonmetal, and the formula of an oxide.
-
ESPN ultimately decided that having Nichols still appear on the network following the NBA Finals was not acceptable. Was this too harsh? Should Nichols have been punished much earlier? Discuss ESPNs...
-
Identify two causes of inflation worldwide and describe whether the inflation from those two causes will abate in the next year. Briefly explain.
-
How does the study of mitochondrial heteroplasmy and its inheritance patterns provide insights into mitochondrial disease variability, and what challenges exist in understanding the clinical...
-
Larry finds a lens and shows it to his friends, Mirez and Sherly. All three look through it. Larry looks at the window across the room with the lens and says the window looks upside down. Mirez said...
-
Pharoah Co. has identified an activity cost pool to which it has allocated estimated overhead of $10212000. It has determined the expected use of cost drivers for that activity to be 851000...
-
How should feedback be solicited and handled following the distribution of a memo? Discuss the process for follow-up communications to ensure understanding and action.
-
There are four shoppers in a store--A, B, C and D--who sequentially show up at two checkout lines: line number 1 and line number 2. Initially, both checkout lines are empty. A shows up first, sees...
-
Suppose Green Network Energy needs to raise money to finance its new manufacturing facility, but their CFO does not think the company is financially capable of making the periodic interest payments...
-
In this problem you need to draw two pictures of solutions in beakers at different points in time. Time zero (t = 0) will be the hypothetical instant at which the reactants dissolve in the solution...
-
You come across a beaker that contains water, aqueous ammonium acetate, and a precipitate of calcium phosphate.
-
Equal quantities of the hypothetical strong acid HX, weak acid HA, and weak base BZ, are added to separate beakers of water, producing the solutions depicted in the drawings. In the drawings, the...
-
s Machine-hours required to support estimated production Fixed manufacturing overhead cost Variable manufacturing overhead cost per machine-hour Required: 1. Compute the plantwide predetermined...
-
How do visionary leaders navigate the complexities of organizational dynamics to cultivate a culture of innovation and adaptability ? Explain
-
Finnish Corporation manufactures custom molds for use in the extrusion industry. The company allocates manufacturing overhead based on machine hours. Selected data for costs incurred for Job 532 are...

Study smarter with the SolutionInn App


