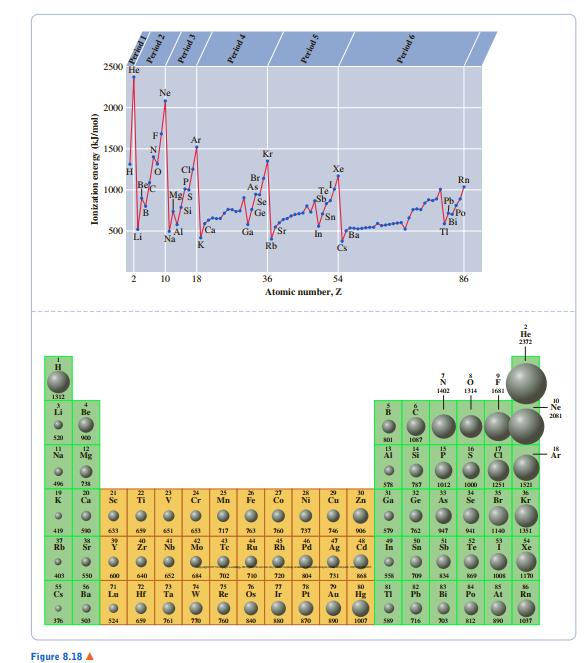
From Figure 8.18, predict the first ionization energy of ununseptium (Z = 117). 2500 He 2000 *
Question:
From Figure 8.18, predict the first ionization energy of ununseptium (Z = 117).
Transcribed Image Text:
2500 He 2000 * 1500 Kr Br As Rn E 1000 Bi S00 Li Ca Sr Na In Ba K Rb Cs 10 18 36 54 86 Atomic number, Z -Ne 吕 550 1008 At 524 730 J0 s40 S0 203 K12 Figure 8.18 A pona Period 2 Period 3 Ionizat ion energy (kJ/mol) Period 4 Perlod 5 つ=2の o 6 Perlod 6 AZ 中 一
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
The ionization energy of ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
From Figure 8.18, predict the first ionization energy of francium (Z = 87). 2500 He 2000 * 1500 Kr Br As Rn E 1000 Bi S00 Li Ca Sr Na In Ba K Rb Cs 10 18 36 54 86 Atomic number, Z -Ne 550 1008 At...
-
The first ionization energy of the chlorine atom is 1251 kJ/mol. Without looking at Figure 8.18, state which of the following values would be the more likely ionization energy for the iodine atom....
-
The first ionization energy of the oxygen molecule is the energy required for the following process: O2 (g) O2+ (g) + e- The energy needed for this process is 1175kJ/mol, very similar to the first...
-
What is a reporting entity?
-
For each of the following, state whether the events created are mutually exclusive and whether they are collectively exhaustive. a. Undergraduate business students were asked whether they were...
-
Following are the accounts and balances from the adjusted trial balance of Stark Company. Prepare the (1) Income statement (2) Statement of owners equity for the year ended December 31 (3) Balance...
-
The weights (in lbs) of 14 newborn babies. Find the range, mean, variance, and standard deviation of the population data set. 7 5 12 12 6 9 11 4 7 6 8 7 10 9
-
The Hatcher Company is in the process of developing a new inventory management system. One of the event handling processes in that system is Receive Supplier Shipments. The (inexperienced) systems...
-
Matthew wants to take out a loan to buy a car. He calculates that he can make repayments of $4000 per year. If he can get a five-year loan with an annual interest rate of 4%, what is the maximum...
-
What specific HR function scan you identify as being problematical in this company? Again, please give specific answers.
-
What property of an atom does nuclear magnetic resonance depend on? What frequency range of the electromagnetic spectrum does NMR use?
-
For eka-lead, predict the electron configuration, whether the element is a metal or nonmetal, and the formula of an oxide.
-
Discuss the advantages and disadvantages of consumer-generated marketing. In only a few short years, consumer-generated marketing has increased exponentially. Its also known as consumer-generated...
-
How does mitochondrial dysfunction contribute to aging and age-related diseases, and what are the proposed mechanisms linking mitochondrial quality, oxidative stress, and cellular senescence ?
-
What is the impact of mitochondrial-nuclear crosstalk on cellular function, and how do defects in the communication between these two genetic systems affect cellular metabolism, stress response, and...
-
Identify a dataset in your domain or interested area (using Git hub or Kaggle based on your desired preference) from the industry and perform a Descriptive Statical analysis to make your dataset a...
-
https://youtu.be/4BZuWrdC-9Q Watch the video with the link that I provided above, explain some strengths and weaknesses of the presentation and what you think you can learn from it.
-
Beth and jerry own a residential home valued at approximately $250,000. while they were away on vacation Lightning struck their house causing a large fire that would approximately $125,000 of damage...
-
Eleven of your friends are about to take a course, and each one is interested in buying one used textbook. Another 11 of your friends have already taken the course and are willing to consider selling...
-
Sue Deliveau opened a software consulting firm that immediately paid $2,000 for a computer. Was this event a transaction for the business?
-
Define the terms strong electrolyte and weak electrolyte. Give an example of each.
-
Part 1 a. Both NaCl and MgCl2 are soluble ionic compounds. Write the balanced chemical equations for these two substances dissolving in water.
-
You need to perform gravimetric analysis of a water sample in order to determine the amount of Ag+ present.
-
- Let [121] A-03 1 4 12 If possible, find a matrix C such that 1 2 AC=01 41
-
Provide References In September of 2015, the EPA issued Volkswagen ( OTCPK:VLKAY ) a notice of violation of the Clean Air Act of 1963. The EPA accused the Volkswagen Group of intentionally...
-
2. Find c such that A is invertible 1 0 A= -1 -1 3 1 0 2c -4]

Study smarter with the SolutionInn App


