What do you expect for the BCl bond length in boron trichloride, BCl 3 , on the
Question:
What do you expect for the B—Cl bond length in boron trichloride, BCl3, on the basis of covalent radii (Table 9.4)?
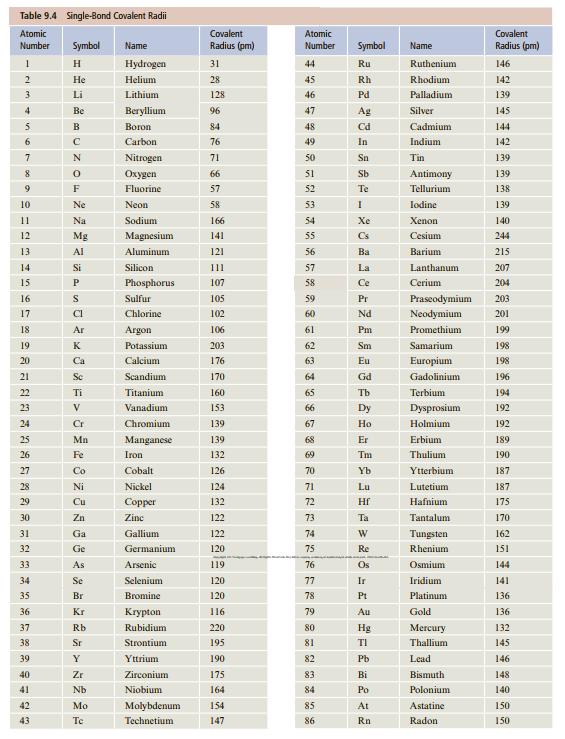
Table 9.4 Single-Bond Covalent Radii Atomic Covalent Atomic Covalent Number Symbol Name Radius (pm) Number Symbol Name Radius (pm) H. Hydrogen 31 44 Ru Ruthenium 146 Не Helium 28 45 Rh Rhodium 142 3 Li Lithium 128 46 Pd Palladium 139 Be Веryllium 96 47 Ag Silver 145 B Вогоn 84 48 Cd Cadmium 144 6. Carbon 76 49 In Indium 142 7 Nitrogen 71 50 Sn Tin 139 8 Охудеn 66 51 Sb Antimony 139 9 F Fluorine 57 52 Te Tellurium 138 10 Ne Neon 58 53 Iodine 139 11 Na Sodium 166 54 Xe Xenon 140 12 Mg Magnesium 141 55 Cs Cesium 244 13 Al Aluminum 121 56 Ba Barium 215 14 Si Silicon 111 57 La Lanthanum 207 15 P. Phosphorus 107 58 Ce Cerium 204 16 Sulfur 105 59 Pr Praseodymium 203 17 CI Chlorine 102 60 Nd Neodymium 201 18 Ar Argon 106 61 Pm Promethium 199 19 K Potassium 203 62 Sm Samarium 198 20 Ca Calcium 176 63 Eu Europium 198 21 Sc Scandium 170 64 Gd Gadolinium 196 22 Ti Titanium 160 65 Tb Terbium 194 23 V Vanadium 153 66 Dy Dysprosium 192 24 Cr Chromium 139 67 Но Holmium 192 25 Mn Manganese 139 68 Er Erbium 189 26 Fe Iron 132 69 Tm Thulium 190 27 Co Cobalt 126 70 Yb Ytterbium 187 28 Ni Nickel 124 71 Lu Lutetium 187 29 Cu Соpper 132 72 Hf Hafnium 175 30 Zn Zine 122 73 Ta Tantalum 170 31 Ga Gallium 122 74 Tungsten 162 32 Ge Germanium 120 75 Re Rhenium 151 33 As Arsenic 119 76 Os Osmium 144 34 Se Selenium 120 77 Ir Iridium 141 35 Br Bromine 120 78 Pt Platinum 136 36 Kr Кгуpton 116 79 Au Gold 136 37 Rb Rubidium 220 80 Hg Mercury 132 38 Sr Strontium 195 81 TI Thallium 145 39 Y Yttrium 190 82 Pb Lead 146 40 Zr Zirconium 175 83 Bi Bismuth 148 41 Nb Niobium 164 84 Po Polonium 140 42 Mo Molybdenum 14 85 At Astatine 150 43 Te Technetium 147 86 Rn Radon 150
Step by Step Answer:
r B 84 pm r ...View the full answer



Related Video
The experiment aims to show the impact of various beverages on teeth by using eggs as a representation of enamel. Three eggs are boiled and then placed in glasses filled with fizzy drinks, vinegar, and mango juice for 24 hours. The shells of eggs are similar to enamel as they are composed of calcium carbonate, and enamel is primarily made of calcium phosphate. The eggs are then observed to demonstrate the effects of the different liquids on teeth and the importance of brushing regularly. The egg placed in fizzy drink has turned dark in color but can be cleaned by brushing with toothpaste and rinsing with water. The egg placed in vinegar has had its shell softened due to the chemical reaction of vinegar and calcium carbonate, which can\'t be reversed. This highlights the fact that acids are more damaging to teeth than other substances. The egg placed in mango juice represents the process of bacteria in the mouth converting sugars and starches into acids that form plaque, which can be prevented by brushing. The use of fluoride in toothpaste is also highlighted as it slows down the demineralization process and protects the enamel. The importance of brushing teeth twice a day is emphasized.
Students also viewed these Sciences questions
-
The combustion of acetylene, C2H2, is a spontaneous reaction given by the equation 2C2H2(g) + 5O2(g) 4CO2(g) + 2H2O(l) As expected for a combustion, the reaction is exothermic. What is the sign of...
-
The air bags that provide protection in autos in the event of an accident expand because of a rapid chemical reaction. From the viewpoint of the chemical reactants as the system, what do you expect...
-
The management of a supermarket wants to find if there is a relationship between the number of times a specific product is promoted on the intercom system in the store and the number of units of that...
-
In Exercises 1114, graph each equation in a rectangular coordinate system. If two functions are indicated, graph both in the same system. Then use your graphs to identify each relations domain and...
-
On November 1, an analyst who has been studying a firm called Computer Sciences believes that the company will make a major announcement before the end of the year. Computer Sciences currently is...
-
Rework Problem 55 assuming that the loan agreement calls for a principal reduction of $14,300 every year instead of equal annual payments. Data from problem 55 Prepare an amortization schedule for a...
-
The following information relates to the business of Restaurant Gigi, and the owner is concerned about the profitability and financial structure of his business at 30 June 2025, especially since the...
-
Hershey's is a familiar name in snacks. There's a good chance you have recently enjoyed one of its products. The company manufactures confectionery products in a variety of packaged forms and markets...
-
Frasier is giving a speech and the power of his sound is 3 . 5 8 mW . What is the sound intensity level 4 . 5 9 m away from Frasier?
-
Raveena is a resident of the Republic. She is 30 years old. She is married in community of property. Her husband did not earn any passive income. The following information relates to the 2021 year of...
-
Calculate the CCl and CC bond lengths in ethyl chloride, C 2 H 5 Cl, using values for the covalent radii from Table 9.4. How do these values compare with the experimental values: CCl, 177; CC, 155...
-
Use covalent radii (Table 9.4) to estimate the length of the PF bond in phosphorus trifluoride, PF 3 . Table 9.4 Single-Bond Covalent Radii Atomic Covalent Atomic Covalent Number Symbol Name Radius...
-
Chooser options. In this problem we denote by \(C\left(t, S_{t}, K, Tight)\), resp. \(P\left(t, S_{t}, K, Tight)\), the price at time \(t\) of the European call, resp. put, option with strike price...
-
K feet above the ground with an initial velocity of 128 feet per second. The height h, in feet, of the rocket above the ground at t seconds after launch is given by the function h(t) = - 16t +...
-
Simplify the expression. 6x8.(-2x6)
-
Simplify. y = 1i95 6
-
During 2005, eBay acquired four different companies. In the schedule below, the acquired companies are listed with the aggregate purchase price and with the estimated acquisition-related costs...
-
The largest athlete at the 2 0 1 2 London Plympic was Judo athlete Ricardo Blas Jr . from Guam. He weighted 4 8 0 . 5 pounds. What was his mass in kilograms?
-
Other things held constant, the higher the price of a good Select one: a. The lower the producer surplus. b. The greater the producer surplus. c. The higher the supply. d. The lower the supply.
-
Consider the function f and its graph. a. Estimate the zeros of the area function b. Estimate the points (if any) at which A has a local maximum or minimum. c. Sketch a graph of A, for 0 x 10,...
-
What is the difference between normal hemoglobin and the hemoglobin associated with sickle-cell anemia? How does this affect the solubility of the hemoglobin?
-
What tends to happen to the hemoglobin in a blood cell of a person who has sickle-cell anemia? How does this affect the shape and oxygen-carrying capacity of the cell?
-
How are phospholipids similar in structure to a soap molecule? What structural difference accounts for the fact that phospholipids form lipid bilayers rather than the micelles that soaps form?
-
Thanasi has a business snowplowing. He uses a 2018 Ford Rapture with a snowplow outfitted to it. While plowing a driveway, he negligently ran it into the side of the neighbour's house. The damage to...
-
Mary was so excited about the holiday shopping season that she could hardly wait for the Black Friday sales. She got up early and spent all day at her favorite store, Mega Deals, shopping. Mary was...
-
This can be done as a group or individually. If done in a group, all group members must submit a copy of this assignment to receive a grade. If you were to open a business, what type of business...

Study smarter with the SolutionInn App


