With the aid of a periodic table (not Figure 9.15), arrange the following in order of increasing
Question:
With the aid of a periodic table (not Figure 9.15), arrange the following in order of increasing electronegativity:
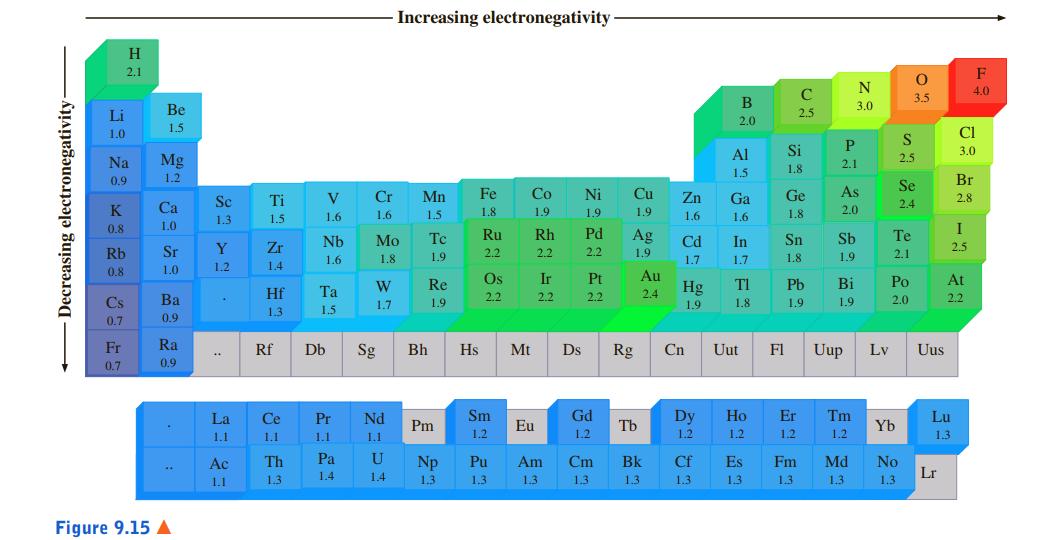
a. Li, Na, Cs
b. B, Be, Li
c. S, Se, Cl
Transcribed Image Text:
Increasing electronegativity H 2.1 F N 4.0 C 3.5 B 3.0 Li Be 2.5 2.0 1.5 1.0 CI Al Si 3.0 Na Mg 2.1 2.5 1.5 1.8 0.9 1.2 Br Se V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Ti 2.4 2.8 K Ca 1.5 2.0 1.6 1.6 1.5 1.8 1.9 1.9 1.9 1.6 1.6 1.8 1.3 1.5 0.8 1.0 I Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te Sr Y Zr 2.5 Rb 1.8 1.9 2.2 2.2 2.2 1.9 1.8 1.9 2.1 1.6 1.7 1.7 0.8 1.0 1.2 1.4 Re Os Ir Pt Au TI Pb Bi Po At Hf Ta Hg Cs Ba 2.2 2.2 2.2 2.4 1.9 2.0 2.2 1.7 1.9 1.9 1.8 1.9 1.3 1.5 0.7 0.9 Fr Ra Rf Db Sg Bh Hs Mt Ds Rg Cn Uut F1 Uup Lv Uus 0.7 0.9 La Ce Pr Nd Sm Gd Dy Ho Er Tm Lu Pm Eu Tb Yb 1.1 1.1 1.1 1.1 1.2 1.2 1.2 1.2 1.2 1.2 1.3 Ac Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No 1.4 1.4 Lr 1.1 1.3 1.3 1,3 1.3 1.3 1.3 1.3 1.3 1.3 1.3 1.3 Figure 9.15 A Decreasing electronegativity Wn
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
a Cs Na Li Electronegativity increases from bottom to ...View the full answer

Answered By

Sumit kumar
Education details:
QUATERNARY Pursuing M.Tech.(2017-2019) in Electronics and Communication Engg. (VLSI DESIGN) from
GNIOT Greater Noida
TERTIARY B.Tech. (2012-2016) in Electronics and Communication Engg. from GLBITM Greater Noida
SECONDARY Senior Secondary School Examination (Class XII) in 2012 from R.S.S.Inter College, Noida
ELEMENTARY Secondary School Examination (Class X) in 2010 from New R.J.C. Public School ,Noida
CERTIFICATION
Summer Training in ‘WIRELESS EMBEDDED SYSTEM’ from ‘XIONEE’ for the six weeks.
EMBEDDED SYSTEM Certificate issued by CETPA INFOTECH for one day workshop.
Certificate of Faculty development program on OPTICAL COMMUNICATION and NETWORKS for one week.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Arrange the following in order of increasing basicity: (a) (b) (c)
-
Arrange the following in order of increasing first ionization energy: Na, Cl, Al, S, and Cs.
-
Arrange the following in order of increasing first ionization energy: F, K, P, Ca, and Ne.
-
In Exercises 1 through 28, differentiate the given function. y = 2x
-
On July 1, a portfolio manager holds $1 million face value of Treasury bonds, the 11 l/4s maturing in about 29 years. The price is 107 14/32. The bond will need to be sold on August 30. The manager...
-
An investment has an installed cost of $527,630. The cash flows over the four-year life of the investment are projected to be $212,200, $243,800, $203,500, and $167,410, respectively. If the discount...
-
You are provided with the following information from the statement of cash flows for Precedent Ltd. Required (a) Calculate the following cash sufficiency ratios for Precedent Ltd for 2025 and 2024:...
-
Jerry Grant, the new controller of Blackburn Company, has reviewed the expected useful lives and salvage values of selected depreciable assets at the beginning of 2010. His findings are as follows....
-
A block slides down a ramp that has a coefficient of kinetic friction of 0.2. The ramp makes an angle of 15 degrees with the horizontal. What is the total force that the block feels if its mass is 3...
-
Compare a companys strategic plan with a football teams game plan.
-
Decide which of the following bonds is least polar on the basis of electronegativities of atoms: ClH, SSi, BrAs.
-
Assuming that the atoms form the normal number of covalent bonds, give the molecular formula of the simplest compound of germanium and fluorine atoms.
-
In Exercises, find the derivative of the function. g(t) = 1 1-2
-
Around the globe (Global Mart) is a dominant force in the international e-commerce landscape, renowned for its extensive product range and vast customer base. As a market leader, the company has...
-
I need the PESTEL ANALYSIS for SAN FRANCISCO CITY, including the aspects: 1. Political 2. Economical 3. Social and cultural 4. Technological 5. Legal Please add the references and bibliography
-
se the Gauss-Jordan reduction to solve the following linear system: X1 X2 + 2x3 == -4 3x1 4x2 + 3x3 -3 -3x1 15x3 = 39 1 X2 X3 000 +
-
How can we relate this back to HR: https://thehustle.co/04242023-trade-school-enrollment/
-
if a private jet flies the same distance in 1 0 hours that a commercial jet flies in five hours. If the speed of the commercial jet was 1 8 3 mph less than three times the speed of the private jet...
-
Would an increase in real GDP and real income in the U.S, other things being equal, cause the dollar to depreciate or appreciate in the foreign exchange market?
-
Use translations to graph f. f(x) = x-/2 +1
-
A solution is made up by dissolving 15.0 g Na2CO310H2O in 100.0 g of water. What is the molality of Na2CO3 in this solution?
-
An aqueous solution is 15.0% by mass of copper(II) sulfate pentahydrate, CuSO45H2O. What is the molarity of CuSO4 in this solution at 20C? The density of this solution at 20C is 1.167 g/mL.
-
An aqueous solution is 20.0% by mass of sodium thiosulfate pentahydrate, Na2S2O35H2O. What is the molarity of Na2S2O3 in this solution at 20C? The density of this solution at 20C is 1.174 g/mL.
-
A horizontal board of negligible thickness and area 4.0 m hangs from a spring scale that reads 60 N when a 2.0 m/s wind moves below the board. The air above the board is stationary. When the wind...
-
The only force acting on a 2.2 kg body as it moves along the positive x axis has an x component Fx = -5x N, where x is in meters. The velocity of the body at x = 2.5 m is 9.0 m/s. (a) What is the...
-
Use conservation of energy to determine the angular speed of the spool shown in the figure below after the 3.00-kg bucket has fallen 3.70 m, starting from rest. The light string attached to the...

Study smarter with the SolutionInn App


