(a) Discuss the factors that contribute towards KCl being a readily soluble salt (35 g per 100...
Question:
(a) Discuss the factors that contribute towards KCl being a readily soluble salt (35 g per 100 g H2O at 298 K).
(b) Develop your answer to part (a) by using the following data: ΔhydHº(K+, g) = −330 kJ mol−1;
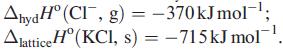
Transcribed Image Text:
AhydH° (CI, g) = -370kJmol-¹; Alattice H (KCl, s) = -715kJ mol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
a Factors contributing towards KCl being a readily soluble salt Ionic nature KCl is an ionic compound consisting of K cations and Cl anions Ionic comp...View the full answer

Answered By

Collins Njuguna
I graduated from Maseno University with a Bachelor of Science in Applied Statistics. After graduation, I started tutoring students in mathematics. My experience in mathematics education is extensive and varied. I have taught a wide range of topics, including algebra, geometry, trigonometry, calculus, statistics, probability, and computer science. I have also worked with students of all ages and backgrounds, from elementary school to college.
My teaching method is based on the idea of hands-on learning. I believe that students learn best when they are actively engaged in the learning process, so I focus on giving students the tools they need to explore the material on their own. I also emphasize the importance of practice and review, as these are essential for mastering math concepts.
I have also developed several online and in-person courses on mathematics. My courses are designed to help students learn mathematics in an efficient and comprehensive way, and I use a variety of activities and exercises to ensure that my students are engaged and motivated.
Overall, my passion for mathematics and teaching has allowed me to be a successful tutor and educator in the field. I am confident that my experience will help your students master the mathematics they need to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Suppose that the person with the above utility function is struck by lightning. He survives, except now his utility function is u = 1 log(x1 1) + 2 log(x2 2). Will his consumption decisions change...
-
The following table gives the systolic blood pressure (SBP), body size (QUET), age (AGE), and smoking history (SMK = 0 if a nonsmoker, SMK = 1 if a current or previous smoker) for a hypothetical...
-
A study was conducted on a sample of 53 patients presenting with prostate cancer who had also undergone a laparotomy to ascertain the extent of nodal involvement (Collett 1991). The result of the...
-
Access various employment Web sites (for example, www.monster.com and www.dice.com ) and find several job descriptions for a database administrator. Are the job descriptions similar? What are the...
-
In the United States, many states and cities deliberately seek investment in their states and communities by firms from other parts of the country. Why are some of those who seek investment from...
-
Elizabeth College, a small private college, had the following transactions in fiscal year 2020. 1. Gross tuition and fees revenue totaled $5,600,000. Tuition waivers and scholarships of $346,000 were...
-
Pileri Industries shipped goods to Consolidated Industries, Inc., via a common carrier. The goods were lost in transit. Pileri claimed that the sale was a shipment contract, thus putting the risk of...
-
Refer to the gasoline sales time series data in Table. a. Compute four- and five-week moving averages for the time series. b. Compute the MSE for the four- and five-week moving average forecasts. c....
-
As part of a survey, 15 adults were asked, "How many hours did you spend at your job last week?" The results are shown in the stem-and-leaf display below. Use the display to answer the questions that...
-
When NaCN dissolves in water, the resulting solution is basic. Account for this observation given that pK a for HCN is 9.31.
-
Potassium chromate is used as an indicator in titrations for the determination of chloride ion. At the end-point of a titration of an aqueous solution of a metal chloride salt (e.g. NaCl) against...
-
Freddys Furnaces, Inc., assigned $550,000 of direct labor costs to production during the current period but paid its employees only $500,000. What are the two journal entries used to record these...
-
Hillshire plc uses a residual dividend policy. (see Question 11.) A debtequity ratio of 0.60 is considered optimal. Earnings for the period just ended were 524,292 and a dividend of 50,000 was...
-
In what situations would you use a holding period return or a percentage return? Are the two measures the same?
-
Mobistar intends to issue callable, perpetual bonds with annual coupon payments. The bonds are callable at 12,500. One-year interest rates are 6 per cent. There is a 60 per cent probability that...
-
When Microsoft launched its Xbox console in 2001 the NPV from the project was negative. However, the company proceeded with the project. Why do you think this was?
-
Since 2007 credit rating agencies have come under a lot of criticism for their role in the subprime mortgage crisis. To what extent do you think credit rating agencies were responsible for the...
-
Explain, in your own words, why there might be a difference between the cash balance per a companys general ledger and the closing balance on its monthly bank statement for the same account.
-
Ball bearings are widely used in industrial applications. You work for an industrial food machinery manufacturer and your role is to design the driveshaft assembly on a new type of equipment that...
-
Use the concept particularly the effects of penetration and shielding on the radial wavefunction, to account for the variation of single-bond covalent radii with position in the periodic table.
-
Discuss the following set of rate constants (k) and activation parameters for water exchange reactions of metal aqua ions. Species [TI(OH)]+ [V(OH)]+ [V(OH)]+ [Cr(OH)]+ [Cr(OH),(OH)]+ [Mn(OH)]+...
-
Given the following mechanism for the formation of a chelate complex, derive the rate law for the formation of the chelate. Discuss the step that is different from that for two monodentate ligands....
-
Many activities are counted in the calculation of a country's GDP. Discuss how each of the following affects the GDP calculation in Canada (8 Marks): A.Agropur Dairy Co-operative in Quebec. (2 Marks)...
-
One that is not perfectly competitive but produces competing price is the monopolistic competition. Under Nash equilibrium, we also have many decision-makers or firms without a single firm dominating...
-
Review your digital portfolio. How will it know the situation that exists before implementing the plan? How will it know the impact of a plan had once implemented? Consider these questions then draft...

Study smarter with the SolutionInn App


