Question: (a) Using the data in Table 1.5, determine a value for H for the process: (b) Comment on the relevance of the sign and magnitude
(a) Using the data in Table 1.5, determine a value for ∆H for the process:
![]()
(b) Comment on the relevance of the sign and magnitude of your answer to part (a) in the light of the fact that many metal oxides with ionic lattices are thermodynamically stable.
Table 1.5
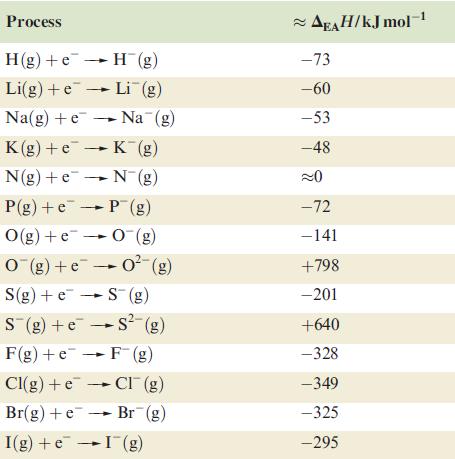
O(g) + 2e 0(g)
Step by Step Solution
3.34 Rating (154 Votes )
There are 3 Steps involved in it
a To determine the value of H for the process Og 2e Og We need to sum the enthalpies of the individual steps involved in the process The enthalpy chan... View full answer

Get step-by-step solutions from verified subject matter experts


