(a) Using the potential diagram below (at pH 14), calculate E O3 2 /O 2 (b) Comment...
Question:
(a) Using the potential diagram below (at pH 14), calculate E°O3−2/O2
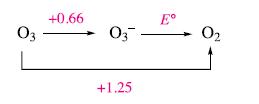
(b) Comment on the following data:
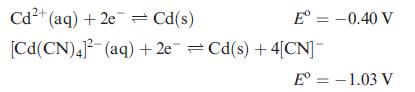
(c) How valid is Fig. 8.4a for aqueous solutions at pH 2?
Figure 8.4a.
Transcribed Image Text:
03 +0.66 03- +1.25 Eº 0₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a To determine For the reaction O 3 2 O 2 O 3 2O 2 we utilize the potential difference between the t...View the full answer

Answered By

Shem Ongek
I am a professional who has the highest levels of self-motivation. Additionally, I am always angled at ensuring that my clients get the best of the quality work possible within the deadline. Additionally, I write high quality business papers, generate quality feedback with more focus being on the accounting analysis. I additionally have helped various students here in the past with their research papers which made them move from the C grade to an A-grade. You can trust me 100% with your work and for sure I will handle your papers as if it were my assignment. That is the kind of professionalism that I swore to operate within. I think when rating the quality of my work, 98% of the students I work for always come back with more work which therefore makes me to be just the right person to handle your paper.
4.80+
174+ Reviews
426+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Carol Harris, Ph.D, CPA, is a single taxpayer and she lives at 674 Yankee Street, Durham, NC 27409. Her Social Security number is 793-52-4335. Carol is an Associate Professor of Accounting at a local...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1 and 2. On September 1, Irene opened a retail store that specializes in sports car...
-
Put all of the below items in the correct format. Don't be concerned about numbers. Just get the lines and headings all in the right places. Beginning Cash Balance CASH FLOWS FROM FINANCING...
-
Of the two methods allied firms can use to combat opportunism, which one do you think is better? Why?
-
You have an IRA worth $200,000 and want to start to make equal, annual withdrawals (i.e., distributions from the account) for 20 years. You anticipate earning 5 percent on the funds. (To facilitate...
-
In 2004, Jui-Chen Lin, a citizen of China, entered into an agreement with Robert Chiu and Charles Cobb, citizens of the United States, to form an LLC to acquire and operate a fast-food restaurant in...
-
John Donelan, the managing partner of the Dublin office of Donelan Consulting, saw the following diagram while visiting one of his clients, who owns a small manufacturing company. Mr. Donelan...
-
Stage duration (hours) for immatures of 2 species of blow fly when reared at several temperatures; e.g., "time to complete" the stage. The threshold temperature for all species is 10C.--------1. What...
-
In hydrochloric acid, HOI reacts to give [ICl 2 ] . Use the potential diagrams below to explain why HOI disproportionates in aqueous acidic solution, but does not when the acid is aqueous HCl. [03]...
-
Use the data in Appendix 11 to rationalize the following observations in a quantitative manner. What assumption(s) have you made in answering this question? (a) The dithionate ion, [S 2 O 6 ] 2 , can...
-
For the following exercises, write the equation of the ellipse in standard form. Then identify the center, vertices, and foci. 9x 2 + y 2 + 54x 4y + 76 = 0
-
The Treadwater Bank wants to raise $1.33 million using three-month commercial paper. The net proceeds to the bank will be $1,304,730. What is the effective annual rate of this financing for...
-
Clovix Corporation has $54.88 million in cash, 9.8 million shares outstanding, and a current share price of $30. Clovix is deciding whether to use the $54.88 million to pay an immediate special...
-
Your company has sales of $103,600 this year and cost of goods sold of $60,300. You forecast sales to increase to $117,700 next year. Using the percent of sales method, forecast next years cost of...
-
Your firm currently has net working capital of $111,000 that it expects to grow at a rate of 5% per year forever. You are considering some suggestions that could slow that growth to 3% per year. If...
-
Your supplier offers terms of 3/20, net 30. What is the effective annual cost of trade credit if you choose to forgo the discount and pay on day 30?
-
Describe the specific identification, FIFO, and weighted- average cost formulas.
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
State Zachariasens two generalizations that favour glass formation and apply them to the observation that cooling molten CaF 2 leads to a crystalline solid whereas cooling molten SiO 2 at a similar...
-
Summarize recent research efforts directed towards finding improved materials for the photocatalytic splitting of water.
-
Explain why the colour of Ta 2 O 5 changes from white to red when it is heated under flowing NH 3 .
-
How did technology affect patterns of urban life in late nineteenth-century America? Under what conditions did the urban poor live? Why did technology fail to help these people?
-
1. Besides federal student loans and private student loans, what are other sources of funding that can help pay for higher education? 2. How do people apply for federal student loans, as well as...
-
The impact of vouchers on the market for private school students can be analyzed through the lens of supply and demand. Initially, the market operates under a pre-voucher equilibrium where the price...

Study smarter with the SolutionInn App


