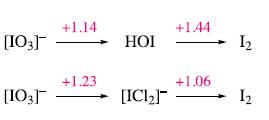
In hydrochloric acid, HOI reacts to give [ICl 2 ] . Use the potential diagrams below
Question:
In hydrochloric acid, HOI reacts to give [ICl2]−. Use the potential diagrams below to explain why HOI disproportionates in aqueous acidic solution, but does not when the acid is aqueous HCl.
Transcribed Image Text:
[03] [03] +1.14 +1.23 НОЇ [ICI2] +1.44 +1.06 Iz Iz
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To understand why HOI disproportionates in aqueous acidic solution but not in aqueous HCl we need to ...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In a laboratory, two liquids, A and B, were found in a box labeled only "isomeric alkyl halides C5H11Br." You have been employed to deduce the structures of these compounds from the following data...
-
When an acid reacts with a base: 1) This is a neutralization reaction 2) Pink color will appear in the resulting solution 3) Both of the answers are correct 4) None of the answers is correct QUESTION...
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
Assume that your team has been in contract with the headquarters of a company that owns several restaurants in different states in the US. Your team is to provide software that manages these...
-
Describe the three most common motives for acquisition.
-
In September, the board of directors of Chaparral Steel approved a 2-for-1 stock split. After the split, how many shares of Chaparral Steel stock will an investor have if she or he owned 400 shares...
-
Panther Racing, LLC, is involved in competitive racing, including the development, maintenance, and operation of a racing team in the IndyCar series. Beginning in 2008, the National Guard annually...
-
Thrifty Markets, Inc., operates three stores in a large metropolitan area. The company's segmented absorption costing income statement for the last quarter is given below: Management is very...
-
For each number-line diagram, list the fractions represented by the arrows. (a) (Use a comma to separate answers as needed. Do not simplify.) (b) (Use a comma to separate answers as needed. Do not...
-
Using data from Appendix 11, and the value for the standard Gibbs energy of formation for PbS of 99 kJ mol 1 , determine a value for K sp for this salt. Data from Appendix 11 The concentration of...
-
(a) Using the potential diagram below (at pH 14), calculate E O3 2 /O 2 (b) Comment on the following data: (c) How valid is Fig. 8.4a for aqueous solutions at pH 2? Figure 8.4a. 03 +0.66 03- +1.25 E 0
-
Do you believe that a businesss carbon footprint will be an important competitive issue in the future?
-
The HNH Corporation will pay a constant dividend of $4 per share, per year, in perpetuity. Assume all investors pay a 25% tax on dividends and that there is no capital gains tax. The cost of capital...
-
EJH Company has a market capitalization of $1.4 billion and 25 million shares outstanding. It plans to distribute $120 million through an open market repurchase. Assuming perfect capital markets: a....
-
FCF Co. has 16,000 shares outstanding and a total market value of $1 million, $350 thousand of which is debt and the other $650 thousand is equity. It is planning a 10% stock dividend. a. What is the...
-
RFC Corp. has announced a $2.30 dividend. If RFCs last price while trading cumdividend is $58, what should its first ex-dividend price be (assuming perfect capital markets)?
-
AMS Company has unexpectedly generated a one-time extra $5 million in cash-flow this year. After announcing the extra cash flow, AMS stock price was $60 per share (it has 1 million shares...
-
Explain when it is appropriate to use each of the three cost formulas.
-
[a] Two foam blocks, each with a charge of 19 micro coulombs (1 C = 10-6 C), are both held in place 19 cm apart in the east-west direction. A foam ball with a charge 49 C is placed 55 cm north of the...
-
The increased reactivity that allows the design and synthesis of materials based on molecular units also means that these compounds are unsuitable for many applications that currently use inorganic...
-
Classify the oxides (a) BeO, (b) TiO 2 , (c) La 2 O 3 , (d) B 2 O 3 , (e) GeO 2 into glass-forming and non-glass-forming.
-
Compare and contrast the chemistries of graphite and C 60 with respect to their compounds in association with the alkali metals.
-
Problem 1(15 points): Use the method of separation of variables to solve the one- dimensional heat equation: - T t x Subject to (1 0
-
A client has indicated an interest in opening a franchise. The client has requested information related to the accounting of operating a franchise. You are to prepare a memo indicating the relevant...
-
Suppose you take the log of the variables in a demand function and estimate the coefficients as noted below: Ln QXD = 5 - 2.2 Ln PX 0.5 Ln I 0.8 Ln A, where, Ln = natural log QXD = quantity demanded...

Study smarter with the SolutionInn App


