Calculate the rate constants for outer-sphere reactions from the following data. Compare your results to the measured
Question:
Calculate the rate constants for outer-sphere reactions from the following data. Compare your results to the measured values in the last column.
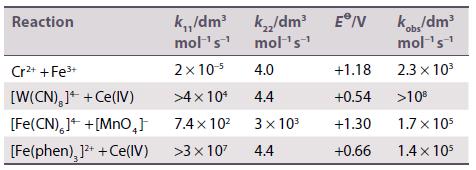
Transcribed Image Text:
Reaction Cr²+ + Fe³+ [W(CN),]++ Ce(IV) [Fe(CN),]+ +[MnO₂] [Fe(phen), ]²+ + Ce(IV) k.,,/dm³ mol ¹s¹ 2x 10-⁹ >4 x 10¹ 7.4×10² >3 x 10² k₂/dm³ E/V mol ¹s¹ 4.0 4.4 3× 10³ 4.4 +1.18 +0.54 +1.30 +0.66 kobs/dm³ mol ¹s¹ 2.3 × 10³ >10⁰ 1.7 x 105 1.4 x 105
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To calculate the rate constants for outersphere reactions we can use the Marcus crossrelation equati...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
BACKGROUND You are an information analyst working for NEE. The company president has asked you to prepare a Quantitative analysis of financial, sales, and operations data to help determine which...
-
The equilibrium Ac=' B + C at 25C is subjected to a temperature jump that slightly increases the concentrations of Band C. The measured relaxation time is 3.0 us. The equilibrium constant for the...
-
Calculate the intrinsic value of Disney by conducting a two-stage dcf company-level valuation analysis, Compare your results to the current market capitalization of the company and Perform a...
-
(5 points) In a study of purchasing behavior at a small shop, it was found that the probabaty that a purchase is more than $5 is 0.29, the probability that a customer will pay with a credit card is...
-
Farley Company identifies the following items for possible inclusion in the taking of a physical inventory. Indicate whether each item should be included or excluded from the inventory taking. (a)...
-
A reversible cycle executed by 1 mol of an ideal gas for which Cp = (5/2)R and Cv = (3/2)R consists of the following: Starting at T1 = 700 K and P1 =1.5 bar. the gas is cooled at constant pressure...
-
Let us consider the scenario tree of Fig. 11.10 and define consider decision Data From Fig. 11.10 Data From Example 11.17 cision variables Z and Z, representing the number of stock shares that we buy...
-
Assume that you were recently hired as assistant to Jerry Lehman, financial VP of Coleman Technologies. Your first task is to estimate Colemans cost of capital. Lehman has provided you with the...
-
Successful career means getting multiple jobs, promotions and earning a handsome salary. Discuss whether you agree with the statement
-
The activation enthalpy for the reduction of cis-[CoCl 2 (en) 2 ] + by Cr 2+ (aq) is 24 kJ mol 1 . Explain the negative value.
-
Octahedral complexes of metal centres with high oxidation numbers or of d metals of the second and third series are less labile than those of low oxidation number and d metals of the first series of...
-
Write the transpose of matrix A. Use the following matrices 1 -2 1 A = 1 B =| 4 1 2 0 1 c- --] -- : 3 4 D = 3 1 E = 5 4 C 1 1. 3 2 1 1 F = -1 1 G = 1 4 3 -4 5 1 Z = 3. 1. 3. 3. 4,
-
Describe the plan for evaluating the needs of remote work during a global pandemic at all four levels of Kirkpatrick's Model. Also provide directions and explanation of how to use the data once...
-
Does Buffet's indicator of valuation help forecast market stock returns.
-
TOPIC : "Impact of Covid on student's mental health" Take sample from australian point of view INTRODUCTION of topic PARTICIPANTS Measures Measure 1 What was the name of the scale and what it...
-
Compare and contrast the benefits and costs of Corporate Social Responsibility for the the automotive industries in various countries. Analyse the potential cultural and institutional factors that...
-
Please send all the _____________ for the Christmas party, out by December 1. Question 19 options: RSVP's RSVP RSVPS RSVPs
-
An emergency rescue vehicle is used an average of 1.3 times daily. Use the Poisson distribution to find a. The probability that the vehicle will be used exactly twice tomorrow b. The probability that...
-
(a) Explain why the concentration of dissolved oxygen in freshwater is an important indicator of the quality of the water. (b) How is the solubility of oxygen in water affected by increasing...
-
If the reaction Fe 2 N(s) + 3/2H 2 (g) 2Fe(s) + NH 3 (g) comes to equilibrium at a total pressure of 1 bar, analysis of the gas shows that at 700. and 800. K, PNH 3 /PH 2 = 2.165 and 1.083,...
-
Draw the structure of an alkyne that can be converted into 3-ethylpentane upon hydrogenation. Provide a systematic name for this compound.
-
Propose a mechanism for each of the following transformations: (a) (b) Na NH3 (1) ,
-
How do institutional power structures shape the dynamics of political decision-making within contemporary societies?
-
1. What are some critical success factors for effectively conducting an HRIS needs analysis? 2. How do companies use an RFP when searching for software? What are the key elements that should be...
-
Perusahaan Otomobil Kedua Sendirian Berhad (English: Second Automobile Manufacturer Private Limited), usually abbreviated as Perodua /prdju/, is the second largest automobile manufacturer in Malaysia...

Study smarter with the SolutionInn App


