Question: Discuss the data presented in Table 12.5. Data from Table 12.5 M+Na+ log K 4.2 K+ 5.9 Rb+ 4.9 2+ Mg+ Ca+ Sr+ Ba+ 2.0
Discuss the data presented in Table 12.5.
Data from Table 12.5
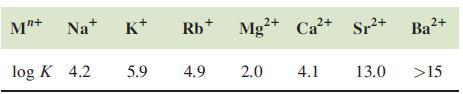
M"+Na+ log K 4.2 K+ 5.9 Rb+ 4.9 2+ Mg+ Ca+ Sr+ Ba+ 2.0 4.1 13.0 >15
Step by Step Solution
3.38 Rating (164 Votes )
There are 3 Steps involved in it
Table 125 presents the log K logarithm of the equilibrium constant values for various metal cations ... View full answer

Get step-by-step solutions from verified subject matter experts


