Discuss the following data for MM and MCO bond enthalpies in a series of metal carbonyl cluster
Question:
Discuss the following data for M−M and M−CO bond enthalpies in a series of metal carbonyl cluster compounds. The enthalpies given in each case are average values for that compound.
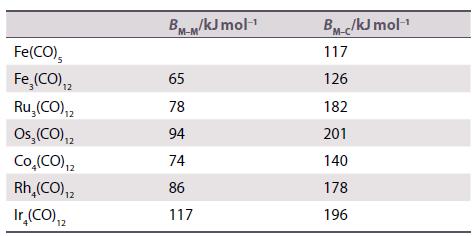
Transcribed Image Text:
Fe(CO), Fe, (CO),₁2 Ru,(CO) 12 Os, (CO) ₁2 Co₂(CO) ₁2 Rh₂(CO) ₁2 Ir₂(CO) ₁2 12 BM-M/kJ mol-¹ 65 78 94 74 86 117 BM-c/kJ mol-¹ 117 126 182 201 140 178 196
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
The data provided shows the average MM metalmetal and MCO metalcarbonyl bond enthalpies in a series of metal carbonyl cluster compounds Lets discuss t...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Suppose that you are given a series of metal tricarbonyl compounds having the respective symmetries C 2v , D 3h , and C s . Without consulting reference material, which of these should display the...
-
The Hydrogenics case is the first in a series of four cases that illustrate a comprehensive analysis of an international corporation. In this case the balance sheet will be analyzed with the income...
-
The State of Virginia has instituted a series of Standards of Learning (SOL) tests in math, history, English, and science that all high school students must pass with a grade of 70 before that are...
-
Write the dual for each of the following primal problems: (a) Maximize z = - 5x1 + 2x2 Subject to - x1 + x2 - 2 2x1 + 3x2 5 X1, x2 0 (b) Minimize z = 6x1 + 3x2 Subject to 6x1 - 3x2 + x3 2 3x1 +...
-
At May 31, Suarez Company has net sales of $330,000 and cost of goods available for sale of $230,000. Compute the estimated cost of the ending inventory , assuming the gross profit rate is 35%.
-
Estimate the density of liquid ethanol at 180(C and 200 bar.
-
The discipline of neuroscience makes use of network theory to identify the structures relating to functionality of the brain. Consider the paper by Vrtes et al. [469]. You do not need to study all...
-
Last Unguaranteed Financial Inc. purchased the following trading securities during 2014, its first year of operations: The market price per share for the trading security portfolio on December 31,...
-
1. Reflect the given shapes over the x-axis and the y-axis. 2. Rotate the given shapes 180. 3. Calculate the area, the surface area, and the volume of the given shapes.
-
The rate of attack on [Co(OH 2 ) 6 ] 3+ by an entering group Y is nearly independent of Y, with the spectacular exception of the rapid reaction with OH . Explain the anomaly. What is the implication...
-
How does each of the following modifications affect the rate of a square-planar complex substitution reaction? (a) Changing a trans ligand from H to Cl ; (b) Changing the leaving group from Cl to...
-
Rewrite the volume for each of the following stocks as sales in 100s. a. Berkshire Hathaway Inc. b. McDonalds Corp c. Verizon d. Toyota
-
Bosworth et al. (2007) state that high quality strategic workforce planning can reduce staff wastage, as well as having other important benefits. Drawing on Mintzbergs theory of strategic planning...
-
In Beckhard's formula for successful change, the V stands for the vision of the desired state. In Beckhard's formula for successful change, the R stands for a realistic approach to the change. Select...
-
) What is the marginal rate of substitution, MRS? State the definition and explain, in words, what it means. b) MRS will have an influence on the shape of an indifference curve. What influence? c)...
-
Write broadcast story (10 lines/30 seconds) only no print for this one based on the information Focus on the news. What's most important in this story? The dead/injured, and the traffic issues. 2....
-
how does Eden demonstrated her knowledge of child development? Layla: 'I'm gunna get some glitter and stick on my fairy.' Cassie: 'Yeah me too. That's a good idea!" Layla: 'No! It's my idea and you...
-
Classify the following random variables according to whether they are discrete or continuous: a. The number of words spelled correctly by a student on a spelling test b. The amount of water flowing...
-
Use integration by parts to evaluate the following. Check your answer by taking the derivative. x2e-xdx
-
A terminal alkyne was treated with NaNH 2 followed by propyl iodide. The resulting internal alkyne was treated with ozone followed by water, giving only one type of carboxylic acid. Provide a...
-
Give an example of two systems that are in equilibrium with respect to only one of two state variables.
-
Aerobic cells metabolize glucose in the respiratory system. This reaction proceeds according to the overall reaction 6O 2 (g) + C 6 H 12 O 6 (s) 6CO 2 (g) + 6H 2 O(l) Calculate the volume of oxygen...
-
You are the CEO of a company that is rapidly expanding globally. You have the opportunity to make a organizational structure that best fits the needs or your organization. How will you structure your...
-
Discuss three ways in which U.S. banks can become involved in international banking. Explain briefly
-
George purchased a rental house (House A") in 2002 for $250,000. While George owned the house, George took depreciation deductions totaling $50,000 (assume this is the proper amount). George also...

Study smarter with the SolutionInn App


