Figure 5.4 shows the formation of three sp 2 hybrid orbitals (see eqs. 5.35.5). (a) Confirm that
Question:
Figure 5.4 shows the formation of three sp2 hybrid orbitals (see eqs. 5.3–5.5).
(a) Confirm that the directionalities of the three hybrids are as specified in the figure.
(b) Show that eqs. 5.3 and 5.5 correspond to normalized wavefunctions.
Equations
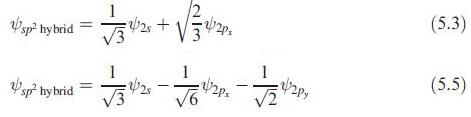
Figure 5.4.
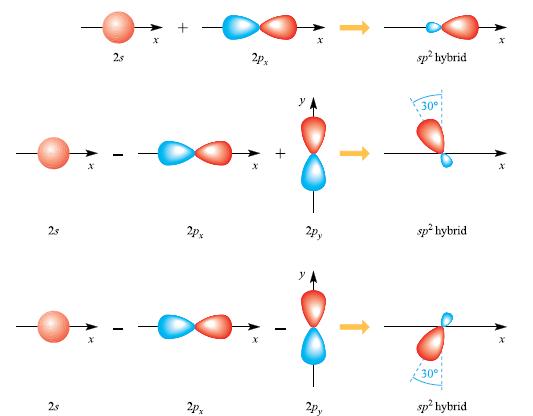
Transcribed Image Text:
sp hybrid 2/25 + 2/20 Psp hybrid 12-12-20 6 2 (5.3) (5.5)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
Lets address each part of the question separately a Confirm that the directionalities of the three hybrids are as specified in the figure In Figure 54 ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
DeArmond Corporation has budgeted sales of 54,000 units, target ending finished goods inventory of 9,000 units, and beginning finished goods inventory of 2,700 units. How many units should be...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The bimolecular reaction of chlorine monoxide can result in the formation of three different combinations of products or product channels (rate constant for each reaction is indicated): Determine the...
-
using System; using System.Collections.Generic; using System.ComponentModel; using System.Data; using System.Drawing; using System.Linq; using System.Text; using System.Threading.Tasks; using...
-
Alecs Designs started business in 2012 with total assets of $25,000 and total liabilities of $20,000. At the end of 2012, Alecs Designs total assets were $65,000, and total liabilities were $12,000....
-
In maize, the genes Pl for purple leaves (dominant over Pl for green leaves), sm for salmon silk (recessive to Sm for yellow silk), and py for pig my plant (recessive to Py for normal-size plant) are...
-
True or False: Multiple roots can exist when using IRR and MIRR methods.
-
John and Jane Darling are newlyweds trying to decide among several available rentals. Alternatives were scored on a scale of 1 to 5 (5 best) against weighted performance criteria, as shown in Table....
-
Direct labor-hours Machine-hours Total fixed manufacturing overhead cost Variable manufacturing overhead per machine-hour Variable manufacturing overhead per direct labor-hour Required: 1. Compute...
-
(a) The structures of cis- and trans-N 2 F 2 were shown in worked example 3.1. Give an appropriate hybridization scheme for the N atoms in each isomer. (b) What hybridization scheme is appropriate...
-
(a) State what is meant by the hybridization of atomic orbitals. (b) Why does VB theory sometimes use hybrid orbital rather than atomic orbital basis sets? (c) Show that eqs. 5.1 and 5.2 correspond...
-
Draw a class inheritance diagram for the following set of classes: Class Goat extends Object and adds an instance variable tail and methods milk( ) and jump( ). Class Pig extends Object and adds an...
-
The 2018 federal poverty guidelines used to determine eligibility for Medicaid and the Childrens Health Insurance Program (CHIP) set a poverty line of $25,100 for a family of four in the 48...
-
As corruption falls in a country, cost of production often falls. Why?
-
Conduct an online search for iPhone repair San Diego You will find about 10 million entries. Now try iPhone screen repair San Diego. You will find more than 6 million entries. Try to estimate the...
-
Most casino game dealers in Las Vegas are paid minimum wage, and therefore rely on tips for a large portion of their income. Within the past few years, a majority of Las Vegas casinos have instituted...
-
If in fact the Chinese government was allocating the rights to export under a quota to the most productive firms, what would you expect to see happen once the quota is lifted?
-
Metro Inc. owns and operates a network of 566 supermarkets and pharmacies in Quebec and Ontario under the Metro, MetroPlus, Super C, Food Basics, Adonis, Brunet, and Pharmacy and Drug Basics banners....
-
5. How much would you need to deposit in an account now in order to have $5,000 in the account in 5 years? Assume the account earns 2% interest compounded monthly. 10. You deposit $300 each month...
-
The paper Atypical compounds of gases which have been called noble (Chem. Soc. Rev., 2007, 36, 1632) provides a thorough account of the range of compounds formed by Group 18 elements. Among the...
-
Draw the Lewis structures of (a) XeOF 4 , (b) XeO 2 F 2 , (c) XeO 6 2 .
-
Identify the xenon compounds A, B, C, D, and E. D HO C xs F Xe F2 2F E A MeBF B
-
The master of coin, Petyr Baelish is offered an investment where interest is calculated according to the following force of interest: 0.02t = 0
-
a more stringent measure of liquidity than the current ratio is referred to as Acid test debt to equity ratio collectability test current asset turnover
-
A taxpayer can face the Civil Fraud Penalty under Section 6663 for all of the following types of tax violations except: A. Concealment or transfer of income B. Failure to make reasonable attempts to...

Study smarter with the SolutionInn App


