Give appropriate bonding descriptions for the aluminiumnitrogen compounds depicted in Fig. 13.26. Figure 13.26. = AIR =
Question:
Give appropriate bonding descriptions for the aluminium–nitrogen compounds depicted in Fig. 13.26.
Figure 13.26.
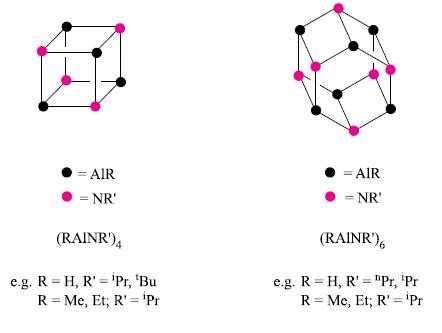
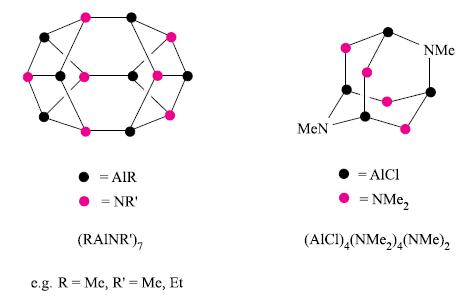
Transcribed Image Text:
= AIR = NR' (RAINR') 4 e.g. R= H, R' = 'Pr, 'Bu R = Me, Et; R' = 'Pr = AIR = NR' (RAINR')6 e.g. R= H, R' = "Pr, 'Pr R = Me, Et; R' = 'Pr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
The following bonding descriptions apply to the aluminumnitrogen compounds shown in Figure 1326 Al...View the full answer

Answered By

Poonam Chaudhary
I have 15 month+ Teaching Experience
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The energy-level diagram in Figure 9.36 shows that the sideways overlap of a pair of p orbitals produces two molecular orbitals, one bonding and one anti-bonding. In ethylene there is a pair of...
-
The IR and mass spectra for three different compounds are shown in Figures 13.45-13.47. Identify each compound. a. b. c. 25 26 2.7 28 19 2000 100 43 58 100 71 85 0 20 40 60 80 100 120 m/z 13 14 15 6...
-
In screening for compounds useful in treating hypertension ( high blood pressure), researchers assign six rats to each of three groups. The rats in group 1 receive .1 mg/ kg of a test compound;...
-
WiCom Servicing completed these transactions during November 2020, its first month of operations: Required 1. Prepare journal entries to record the transactions. Use general journal page 1. 2. Set up...
-
The trustees of the Danube School of Art and Music, located in Tuttlingen, Germany, are considering a major overhaul of the schools audio system. With or without the overhaul, the system will be...
-
The dataset FloridaLakes includes information on lake water in Florida. We want to build a model to predict AvgMercury, which is the average mercury level of fish in the lake. Start with a model...
-
A university pumps its water from wells located on campus. The falling water table has caused pumping costs to increase, the quantity of water available to decrease, and the quality of water to...
-
The following are activities that occurred at Franklin Manufacturing, a nonpublic company. 1. Franklin's accountant did not record checks written in the last few days of the year until the next...
-
Suppose that T: R R is a linear map, and . . . 2 2 3 T2 3 T1 = 2 and T 0 2 5 0 Then 2 T4 7
-
(a) Write down, in order, the names and symbols of the elements in group 13; check your answer by reference to the first page of this chapter. (b) Classify the elements in terms of metallic and...
-
Suggest likely products for the following reactions: (a) BCl3 + EtOH (b) BF3 + EtOH (c) BCl3 + PhNH (d) BF3 +KF-
-
Oxygen at 100 kPa and 200oC is compressed to half its initial volume. Determine the final state in terms of pressure (p2) and temperature (T2) if the compression is carried out in an (a) Isobaric....
-
Siemens is a global electronics and electrical engineering corporation, operating in energy, healthcare, infrastructure and industrial solutions sectors. Founded in Germany in 1925, the company has...
-
Making HRM decisions is often a group activity, as seldom does one person have all the required information, context, and expertise to tackle every HR issue. Working in a group, review the six trends...
-
The city of Jasper has conducted a job analysis of its firefighters. The job being analyzed is a fire apparatus operator/driver (FAOD). In addition to conducting regular firefighting activities,...
-
A retaining wall is shown in Figure 14.36. For each problem, determine the Rankine active force, Pa, per unit length of the wall and the location of the resultant. H H, Y V2 12 m 4 m 17 kN/m 23.2...
-
A test vendor has approached the vice president of HR in your company with a new test of conscientiousness. The test vendor claims that the new conscientiousness test is at least as valid as the old...
-
On September 1, 2013, Franklin Inc. acquired 40% of the voting shares of Gould Ltd. Under the companys constitution, each share is entitled to one vote. On the basis of past experience, only 65% of...
-
Find the reduced echelon form of each of the matrices given in Problems 120. c 1 26 + 4
-
Use the Kapustinskii equation and the ionic and thermochemical radii given in Resource section 1 and Table 4.10, and r(Bk 4+ ) = 96 pm to calculate lattice enthalpies of (a) BkO 2 , (b) K 2 SiF 6 ,...
-
On the basis of the factors that contribute to lattice enthalpies, place LiF, SrO, RbCl, AlP, NiO, and CsI, all of which adopt the rock-salt structure, in order of increasing lattice enthalpy.
-
Explain, by considering the magnitude of the key terms in the BornHaber cycle that involve the halogen or halide ion, why AgF 2 exists but AgCl 2 does not.
-
The Nad Company manufactures and sells the XR chair. It pays $1900 per month for rent and utilities, and $4500 per month for management salaries. The variable costs are $95 per unit and each unit...
-
Use the following information to answer this question. Windswept, Incorporated 2022 Income Statement (in millions) Net sales Cost of goods sold Depreciation 7:23 Earnings before interest and taxes...
-
Now that you have learned the basics of using the ASC database, it is time for you to practice actually finding GAAP to answer an accounting question. As an accounting professional, you will use the...

Study smarter with the SolutionInn App


