In Fig. 6.11, which of the boundaries depend on the choice of Fe 2+ concentration as 10
Question:
In Fig. 6.11, which of the boundaries depend on the choice of Fe2+ concentration as 10−5 mol dm−3?
Figure 6.11.
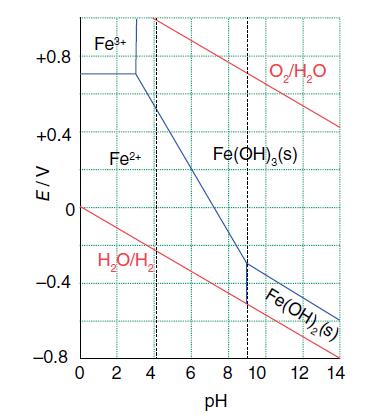
Transcribed Image Text:
+0.8 +0.4 E/V -0.4 -0.8 Fe3+ Fe2+ H,O/H, 024 Fe(OH),(s) Fe(OH), (s) 6 8 10 12 14 pH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
In the given table the boundaries that depend on the choice of Fe2 concentration as 105 mol dm3 are ...View the full answer

Answered By

Chandrasekhar Karri
I have tutored students in accounting at the high school and college levels. I have developed strong teaching methods, which allow me to effectively explain complex accounting concepts to students. Additionally, I am committed to helping students reach their academic goals and providing them with the necessary tools to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Assuming that Ska Companys cost of equity capital is 14% and it expects to grow earnings at a rate of 8% per year, we would expect Skas P/E ratio to be ?
-
Presidio Manufacturing is preparing its master budget for the first quarter of the upcoming year. The following data pertain to Presidio Manufacturing's operations: Current assets as of December 31...
-
Fill the blanks of the following table (last column) and find out the value of SDR in terms of U.S. dollars ($/SDR) and the value of U.S. dollars in terms of SDR (SDR/$) The exchange rate is...
-
For each category, indicate which condition is associated with higher rivalry amongcompetitors. Number of firms Fixed costs Level of product High High High Low Low Low differentiation Industry growth...
-
Eleven percent of U.S. adults eat fast food four to six times per week. You randomly select 12 U.S. adults. Find the probability that the number of U.S. adults who eat fast food four to six times per...
-
How many times can you define a function?
-
Listed below are items that are commonly accounted for differently for financial reporting purposes than they are for tax purposes. Instructions For each item below, indicate whether it involves: 1....
-
Executives at Southwestern Construction have noticed that the company's construction team in the Phoenix office is more efficient with its resources than the other teams in the Las Vegas, Salt Lake,...
-
Consult the Ellingham diagram in Fig. 6.16 and determine if there are any conditions under which aluminium might be expected to reduce MgO. Comment on these conditions. Figure 6.16. A,G*/ (kJ md-)...
-
Adding NaOH to an aqueous solution containing Ni 2+ results in precipitation of Ni(OH) 2 . The standard potential for the Ni 2+ /Ni couple is 0.25 V and the solubility product K sp = [Ni 2+ ] [OH ]...
-
Some individuals have indicated that the IASB must be cognizant of the economic consequences of its pronouncements. What is meant by "economic consequences"? What dangers exist if politics play too...
-
I have the following method, and I need to review a tests methods; however, when running coverage gives me a warning one of the two branches missed here && gender == that.gender && Objects. equals...
-
The predictive power of 52-week high in future stock returns. B. Analysts are slow to update their opinions of a stock after new fundamental information has been released. C. Investors increase their...
-
2. Identify the premium, deductible, and co-pay for a Bronze, Silver, Gold, and Platinum plan. Plan Co-Pay/Co-insurance Deductible Premium Bronze 1,500 7,500 365.36 Silver 3,100 5,800 499.51 Gold...
-
D Question 6 What does Kappa statistic or Cohen's Kappa measure? 3 points 3 pts
-
Complete the following exercises by the due date. Make sure your source code using appropriate programming style (e.g., descriptive variable names, indenting, comments, etc.). Exercises Programming...
-
Consider the galvanic cell based on the following halfreactions: b. Calculate ÎGo and K for the cell reaction at 25oC. c. Calculate cell at 25oC when [Au3+] = 1.0 Ã 102 M and [Tl+] = 1.0...
-
Nate prepares slides for his microscope. In 1 day he prepared 12 different slides. Which equation best represents y, the total number of slides Nate prepares in x days if he continues at this rate? A...
-
Ascorbate oxidase contains four copper centres. Discuss their coordination environments, and classify the centres as Type 1, 2 or 3. What is the function of ascorbate oxidase and how do the copper...
-
Compound 29.33, H 4 L, is a model for the siderophore desferrioxamine. It binds Fe 3+ to give the complex [Fe(HL)]. What features does 29.33 have in common with desferrioxamine? Suggest a reason for...
-
Describe the structure of the copper site in plastocyanin and discuss the features of both the metal centre and metal-binding site that allow it to function as an electron-transfer site.
-
As shown in Figure 2, for the given program, c) Draw the Traditional Pipeline. d) Draw the RISC Pipeline with inserted NOOP. e) Draw the RISC Pipeline with reversed instructions. Address Code Block...
-
6.10 Given the SR flip-flop of Fig. P6.10a, complete the timing diagram of Fig. P6.10b by determining the waveform of the output Q. Note that the flip-flop is triggered on the positive edge of the...
-
9. What are the preorder, inorder, and postorder traversals of the binary tree shown below? M)

Study smarter with the SolutionInn App


