One member of each of the following sets of compounds is not isoelectronic with the others. Which
Question:
One member of each of the following sets of compounds is not isoelectronic with the others. Which one in each set is the odd one out?
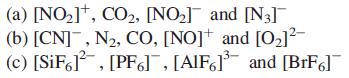
Transcribed Image Text:
(a) [NO₂], CO₂, [NO₂] and [N3] (b) [CN], N₂, CO, [NO] and [0₂]²- (c) [SiF6], [PF6], [AlF6]³ and [BrF]¯¯
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To identify the odd one out in each set of compounds based on isoelectronic species we need to compa...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Googles ease of use and superior search results have propelled the search engine to its num- ber one status, ousting the early dominance of competitors such as WebCrawler and Infos- eek. Even later...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
2. Evaluate the following definite integrals (a) (2x - 6x) dx /6 (b) 16 cos 3t + 2 sin 3t dt x+1 2 da
-
Liberty Advertising, Inc., engaged in the following business transactions during July of 2012: Jul 1 Borrowed $325,000 from Oakville Bank. The company president signed a note payable to the bank in...
-
Explain why pay comparability legislation requiring that the public sector remunerate government employees at wages equal to private sector counterparts might create excess supplies of labor in...
-
Show that after nearly all of the positrons were annihilated and the electron number density had nearly leveled off at the proton density, the ratio of the positron number density to the photon...
-
McGill and Smyth have capital balances on January 1 of $50,000 and $40,000, respectively. The partnership income-sharing agreement provides for (1) annual salaries of $22,000 for McGill and $13,000...
-
A 47-kg pole vaulter running at 10 m/s vaults over the bar. Her speed when she is above the bar is 1.5 m/s. Neglect air resistance, as well as any energy absorbed by the pole, and determine her...
-
Using the data in Table 2.2, determine which of the following covalent single bonds is polar and (if appropriate) in which direction the dipole moment acts. (a) NH; (b) FBr; (c) CH; (d) PCl; (e) NBr....
-
Draw Lewis structures to describe the bonding in the following molecules: (a) F 2 ; (b) BF 3 ; (c) NH 3 ; (d) H 2 Se; (e) H 2 O 2 ; (f) BeCl 2 ; (g) SiH 4 ; (h) PF 5 .
-
Can audit managers on the audit engagement team, who are also attorneys admitted to the state bar, assist in the defense of a lawsuit against an audit client for product liability defects?
-
What is a budget deficit, and what are the two main ways in which the government can finance deficit spending? Which of these methods of financing deficits does the U.S. government most commonly use?
-
How does the Taylor rule relate to the monetary policy curve?
-
How did financial innovations in mortgage markets contribute to the 20072009 financial crisis?
-
Starting from a situation of long-run equilibrium, what are the short- and long-run effects of a positive demand shock?
-
Describe the effects on the economy if the Federal Reserve uses monetary policy to burst a wrongfully identified asset-price bubble.
-
Identify and discuss some of the common reasons companies lease capital assets.
-
Cornell and Roberts are partners who agree to admit Stanley to their partnership. Cornell has a capital balance of $80,000 and Roberts has a capital balance of $120,000. Cornell and Roberts share net...
-
In their article A thermochemical study of ceria: exploiting an old material for new modes of energy conversion and CO 2 mitigation (Philos. Trans. R. Soc. London, 2010, 368, 3269), Chueh and Haile...
-
Using the following aqueous acid solution reduction potentials E (Pd 2+ ,Pd) = +0.915 V and E ([PdCl 4 ] 2 ,Pd) = +0.60 V, calculate the equilibrium constant for the reaction Pd+ (aq) + 4 Cl(aq)...
-
Draw a Frost diagram for mercury in acid solution, given the following Latimer diagram: Comment on the tendency of any of the species to act as an oxidizing agent, a reducing agent, or to undergo...
-
Draw a state machine diagram that will depict the object states that an object of a Circle class can be in. These statuses are important for reporting, decision making and to easily identify those...
-
Are there any other types of innovations that Tesla achieved other than 'business model innovation'? Identify the type of innovations and explain your rationale.
-
Global Enterprises is a multinational corporation with operations on four continents. The CEO's direct reports include vice presidents for the North American, Asian. African and European divisions....

Study smarter with the SolutionInn App


