Suggest likely products for the following reactions (which are balanced on the left-hand sides) in liquid NH
Question:
Suggest likely products for the following reactions (which are balanced on the left-hand sides) in liquid NH3.
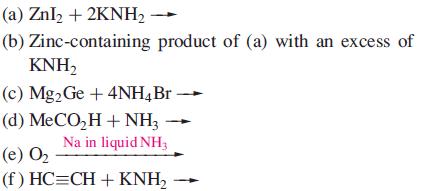
How does reaction (d) differ from the behaviour of MeCO2H in aqueous solution?
Transcribed Image Text:
(a) ZnI₂ + 2KNH₂ - (b) Zinc-containing product of (a) with an excess of KNH₂ (c) Mg2 Ge + 4NH4Br (d) (e) 0₂ MeCO,H+NH, Na in liquid NH3 (f)HC=CH+KNH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
To suggest likely products for the given reactions in liquid ammonia NH3 we should consider the solvents unique properties Liquid ammonia is a powerfu...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For the following reactions at constant pressure, predict if H . E, H , E, or H = E. a. 2HF(g) H2(g) + F2(g) b. N2(g) + 3H2(g) 2NH3(g) c. 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(g)
-
On October 15, 2017, Oil Products Co. purchased 4,000 barrels of fuel oil with a cost of $240,000 ($60 per barrel). Oil Products is holding this inventory in anticipation of the winter 2018 heating...
-
On October 15, 2019, Oil Products plc purchased 4,000 barrels of fuel oil with a cost of £240,000 (£60 per barrel). Oil Products is holding this inventory in anticipation of the winter...
-
Suppose you have a list of blood platelet counts from 500 patients in a hospital. Which of the following is most helpful in understanding the distribution of those values: frequency table, pie chart,...
-
McKeag Corporation manufactures agricultural machinery. At a recent staff meeting, the following direct-labor variance report for the year just ended was presented by the controller. McKeags...
-
Explain how networking and/or negotiating affected behavior, human relations, and performance where you work(ed).
-
Mr. Tam is the front man for the Asian American dance-rock band The Slants. Mr. Tam named his band The Slants to reclaim and take ownership of Asian stereotypes. The band draws inspiration for its...
-
Carey Company had sales in 2016 of $1,560,000 on 60,000 units. Variable costs totaled $900,000, and fixed costs totaled $500,000. A new raw material is available that will decrease the variable costs...
-
explain diagram of ER model and relational model and attach their diagrams too Each cinema is identified by its name and has its residency at an address which consists of a street and city only....
-
(a) In aqueous solution at pH 0, Mn 3+ disproportionates to MnO 2 and Mn 2+ . Write equations for the two half-reactions involved in this process. (b) Use Fig. 8.2 to obtain values of E for the...
-
Use the data in Appendix 11 to rationalize quantitatively why: (a) Mg liberates H 2 from dilute HCl, but Cu does not; (b) Br 2 liberates I 2 from aqueous KI solution, but does not liberate Cl 2 from...
-
If a person saves $63 a month by using coupons and doing comparison shopping, (a) What is the amount for a year? (b) What would be the future value of this annual amount over 10 years, assuming an...
-
Betty owns a farm. On the land are a barn and other farm buildings. Under the surface of the land are valuable minerals. Bettys deed does not indicate any significant limits on her rights to the...
-
Dan owns a half acre of land that fronts on Blue Lake. Rod owns the property behind Dans land. No road runs to Dans land, but Rods driveway runs between a road and Dans property, so Dan uses Rods...
-
Carmen Johnson operated CJ Lending as a so-called credit-repair business. Real estate agent Edgar Tibakweitira paid Johnson to fabricate credit histories for certain individuals who had no such...
-
Dave owns an office building. Dave sells the building to P&I Corporation. To be valid, the deed that conveys the property from Dave to P&I must include a description of the property and a. only Daves...
-
Michelle and Joseph Ardito owned two horses. At one point in time, both Michelle and Joseph were incapacitated. They agreed to have the horses transferred to the Woodbridge Animal Services Farm....
-
Manitoba Exporters Inc. (MEI) sells Inuit carvings to countries throughout the world. On December 1, Year 5, MEI sold 10,000 carvings to a wholesaler in a foreign country at a total cost of 600,000...
-
l ask this second time correnct answer is 38,01 can we look pls Consider a non-conducting rod of length 8.8 m having a uniform charge density 4.5 nC/m. Find the electric potential at P, a...
-
H 3 PO 4 , H 3 PO 3 , and H 3 PO 2 all have a pK a value of 2, but the pK a values of HOCl, HClO 2 , and HClO 3 are 7.5, 2.0, and 3.0, respectively. Explain this observation.
-
The aqueous solution pK a values for HOCN, H 2 NCN, and CH 3 CN are approximately 4, 10.5, and 20 (estimated), respectively. Explain the trend in these cyano derivatives of binary acids and compare...
-
Using hardsoft concepts, which of the following reactions are predicted to have an equilibrium constant greater than 1? Unless otherwise stated, assume gas-phase or hydrocarbon solution and 25C. (a)...
-
Ajax Corp's sales last year were $460,000, its operating costs were $362,500, and its interest charges were $12,500. What was the firm's times-interest-earned (TIE) ratio?
-
Nonconstant growth Microtech Corporation is expanding rapidly and currently needs to retain all of its earnings; hence, it does not pay dividends. However, investors expect Microtech to begin paying...
-
BBB Co. is a 7% Value Added Tax (VAT) registered company and use periodic inventory system. The company distributes computers to retail stores. During the month of August, the following merchandising...

Study smarter with the SolutionInn App


