Suggest possible products for the following reactions: (a) (c) (d) OcFe Oc OCTi. oc I
Question:
Suggest possible products for the following reactions:
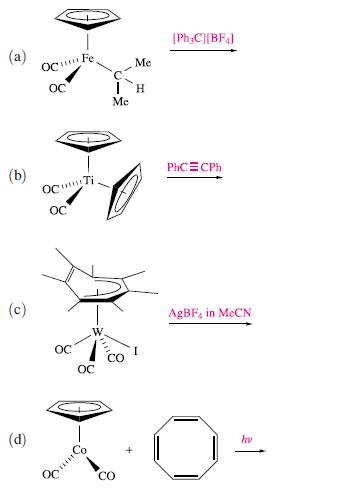
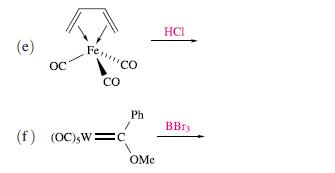
Transcribed Image Text:
(a) (c) (d) OcFe Oc OCTi. oc ос ос I H Me Me CO [PhzC][BF4] PhC=CPh AgBF4 in McCN hu
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 20% (5 reviews)
The reactions presented involve various organometallic complexes and reagents which lead to different types of chemical transformations Here are the p...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Suggest products for the following reactions (which are not balanced): (a) AgCI+ CIF3 (b) CIF + BF3 (c) CsF+IFs - (d) SbF5 + CIF5 (e) Me NF + IF A (f) K[BrF4]
-
Suggest products for the following reactions (which are not necessarily balanced on the left-hand sides): (a) CsF+ XeF4 (b) SiO + XeOF4 (c) XeF + SbF5 (d) XeF6 + [OH] (e) KrF + HO-
-
Suggest likely products for the following reactions (which are balanced on the left-hand sides) in liquid NH 3 . How does reaction (d) differ from the behaviour of MeCO 2 H in aqueous solution? (a)...
-
In December 2014, Shire Computer's management establishes the 2015 predetermined overhead rate based on direct labor cost. The information used in setting this rate includes estimates that the...
-
Using these data from the comparative balance sheet of Ramirez Company, perform horizontalanalysis. December 31, 2014 Accounts receivable Inventory Total assets 460,000 780,000 3,164,000 December 31,...
-
In problem, form a polynomial function whose real zeros and degree are given. Zeros: -1, multiplicity 1; 3, multiplicity 2; degree 3
-
Respondents Varsity Brands, Inc., Varsity Spirit Corporation, and Varsity Spirit Fashions & Supplies, Inc., design, make, and sell cheerleading uniforms. Respondents have obtained or acquired more...
-
The manager of a utility company in the Texas panhandle wants to develop quarterly forecasts of power loads for the next year. The power loads are seasonal, and the data on the quarterly loads in...
-
1. Find the mass of 3.02 x 10 19 atoms of arsenic, which has a molar mass of 74.9 g/mol. 2. Gold has a molar mass of 197 g/mol. (a) How many moles of gold are in a 3.03 g sample of pure gold? (b) How...
-
Illustrate, with examples, what is meant by (a) Oxidative addition, (b) Reductive elimination, (c) -hydrogen abstraction, (d) -hydrogen elimination, (e) Alkyl migration (f) Orthometallation.
-
Suggest products in the following reactions, and give likely structures for the products: (a) Fe(CO) 5 irradiated with C 2 H 4 ; (b) Re 2 (CO) 10 with Na/Hg; (c) Na[Mn(CO) 5 ] with ONCl; (d)...
-
Would you ever pay out dividends when your firms annual net profit is negative? Why? What effect could this have on a firms strategies?
-
A steel wide flange beam has the dimensions shown in the figure. If it is subjected to a shear of V=80 kN, what is the shear stress at B, B' and C locations? B' B 25.2MPa, 25.2MPa, 25.2MPa 1.13MPa,...
-
A storage tank contains a liquid at depth y, where y = 0 when the tank is half full. Liquid is withdrawn at a rate that depends on the depth of the liquid. The contents are resupplied at a sinusoidal...
-
A water molecule and a neutral carbon atom are initially 6.0 10 -6 m apart (about 60000 atomic diameters), and there are no other particles in the vicinity. The polarizability of a carbon atom has...
-
Function f is defined by (x)=2x+c. If (6) = 1, what is the value of f(c)?
-
As shown in the figure, a ball with a mass of 0.420 g and positive charge of q=37.1 C is suspended on a string of negligible mass in a uniform electric field. 4 8 mg Uniform E-field We observe that...
-
Jonathan, a physician, earns $200,000 from his practice. He also receives $18,000 in dividends and interest from various portfolio investments. During the year, he pays $45,000 to acquire a 20%...
-
Funds are separate fiscal and accounting entities, each with its own self-balancing set of accounts. The newly established Society for Ethical Teachings maintains two funds-a general fund for...
-
Propose a method for achieving the following transformation:
-
Predict the product for each of the following electrocyclic reactions: (a) (b) (c) (d) eat hv
-
Predict which side of the following equilibrium is favored, and explain your choice. Heat
-
Ms. Kim had employment-related travel expenses as follows: Meals $1,300 Hotels 3,500 Total $4,800 Assume that the meals were incurred while travelling away from her employer's place of business for...
-
Fin Company maintains a perpetual inventory system and uses the weighted average method of assigning costs. Purchases and sales of inventory for the month of September are as follows: Date Activities...
-
In the e-learning module, there was a discussion about direct costs, indirect costs, and intangible costs. After reading the article on the Ebola epidemic, answer the following: a. What do you think...

Study smarter with the SolutionInn App


