Suggest products for the following reactions; the equations are not necessarily balanced on the left hand sides.
Question:
Suggest products for the following reactions; the equations are not necessarily balanced on the left hand sides. Draw the structures of the sulfur containing products.
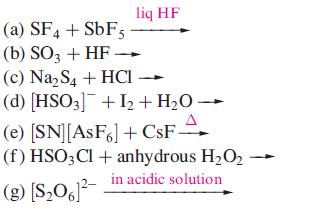
Transcribed Image Text:
(a) SF4 + SbF5 (b) SO3 + HF. liq HF (c) Na₂S4 + HCI (d) [HSO3] + I₂ + H₂O → (e) [SN] [AsF6] + CsF- (f) HSO3Cl + anhydrous H₂O2 in acidic solution (g) [$₂061²-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
lets look at the products of the given reactions involving sulfurcontaining compounds a...View the full answer

Answered By

Churchil Mino
I have been a tutor for 2 years and have experience working with students of all ages and abilities. I am comfortable working with students one-on-one or in small groups, and am able to adapt my teaching style to meet the needs of each individual. I am patient and supportive, and my goal is to help my students succeed.
I have a strong background in math and science, and have tutored students in these subjects at all levels, from elementary school to college. I have also helped students prepare for standardized tests such as the SAT and ACT. In addition to academic tutoring, I have also worked as a swim coach and a camp counselor, and have experience working with children with special needs.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Suggest products for the following reactions; the equations are not necessarily balanced on the lefthand sides. (a) Pl3 + IBr + GaBr3 (b) POBr3 + HF + AsF5 A (c) Pb(NO3)2 liquid NH3 (d) PH3 + K (e)...
-
Suggest products for the following reactions (which are not necessarily balanced on the left-hand sides): (a) CsF+ XeF4 (b) SiO + XeOF4 (c) XeF + SbF5 (d) XeF6 + [OH] (e) KrF + HO-
-
Suggest products and write balanced equations for each of the following reactions; these are not necessarily balanced on the left-hand side. (a) KOH + HSO4 (b) NaOH + SO (c) KOH + CH5OH (d) Na +...
-
What type of isomers are exhibited by [Fe(en) 3 ]Cl 2 (en = ethane-1,2-diamine)? no isomers are possible. cis and trans isomers fac and mer isomers optical isomers
-
Company P, a U.S. company, has a foreign subsidiary in Country Q, where various forms of bribery are accepted and expected. Company P sent one of its top U.S. managers to oversee operations in its...
-
What is malware?
-
Consider the calculation of an external rate of return (ERR). The positive cash flows in the cash flow profile are moved forward to \(t=n\) using what value of \(i\) in the \((F \mid P i, n-t)\)...
-
Presented below are the trial balance and the other information related to Yorkis Perez, a consulting engineer. 1. Fees received in advance from clients $6,000.2. Services performed for clients that...
-
Explore the procedure for ethical decision-making in the face of conflicting obligations, as described in Chapter 2. List the various conflicting obligations that Goldman Sachs executives might have...
-
(a) How many degrees of vibrational freedom does each of ClF 3 and BF 3 possess? The IR spectrum of ClF 3 in an argon matrix exhibits six absorptions, whereas that of BF 3 has only three. Explain why...
-
(a) Comment on the fact that HOI disproportionates in aqueous solution at pH 0, but in aqueous HCl at pH 0, iodine(I) is stable with respect to disproportionation. (b) The solid state structure of...
-
What types of cash flows does a bond buyer receive?
-
The following table shows cash flow data for Rocket Transport. Cash dividend Purchase of bus Interest paid on debt Sales of old equipment Repurchase of stock Cash payments to suppliers Cash...
-
For the following state table, design the system using T flip flops: Q A B C X = 0 A B A X = 1 B C Show the equations and k-maps for z, T1, and T2. Z 1 1 0
-
Find gcd(748,2024) using Euclidean algorithm.
-
Fill the Address Symbols Table after the 2nd pass of the assembler. D1, D2, SH4, ORG LDA STA STA 29 VAL OP1 OP2 SH4 ABCD BSA HEX DEC -4 STA HLT DEC LDA ISZ BUN ORG ANS -2 SH4 SH4 SH4 8F Assume each...
-
ancial Reporting and Analysis X PDF al%20Reporting%20and%20Analysis%20Supplementary%20OSA.pdf Financial Reporting and Analysis X a Ask Copilot + Balances in the ledger as at 28 February 2023 Capital:...
-
In the early 1990s researchers at The Ohio State University studied consumer ratings of six fast-food restaurants: Borden Burger, Hardees, Burger King, McDonalds, Wendys, and White Castle. Each of...
-
Test whether the 5-year survival rate for breast cancer is significantly different between African American and Caucasian women who are younger than 50 years of age and have localized disease....
-
Heating a complex oxide under ammonia can lead to nitridation (with nitrogen replacing oxygen) or reduction (with the production of nitrogen and water). Describe possible products that might be...
-
When NiO is doped with small quantities of Li 2 O, the electronic conductivity of the solid increases. Provide a plausible chemical explanation for this observation.
-
To obtain high oxidation states for first row d metals in complex oxides, e.g. Sr 2 Fe(IV)O 4 , compounds are normally prepared at as low a temperature as possible commensurate with the reaction....
-
Access DRL's annual report and 20-F for the 2019-20 fiscal year on the Investor Relations section of the company's website (www.drreddys.com/investors/reports-and-filings). Answer the following...
-
Describe Kroger's accumulated earnings for each of the 2 years and the meaning of the accumulated earnings account on the balance sheet. Determine the par value of the common shares based on Kroger's...
-
If you were the accountant at your current (or former) place of employment, would you prefer to use static or flexible budgeting? Explain your rationale and the factors that drive your choice. Your...

Study smarter with the SolutionInn App


