The EI mass spectrum of lead(II) acetate shows four peak envelopes, each with an isotope pattern characteristic
Question:
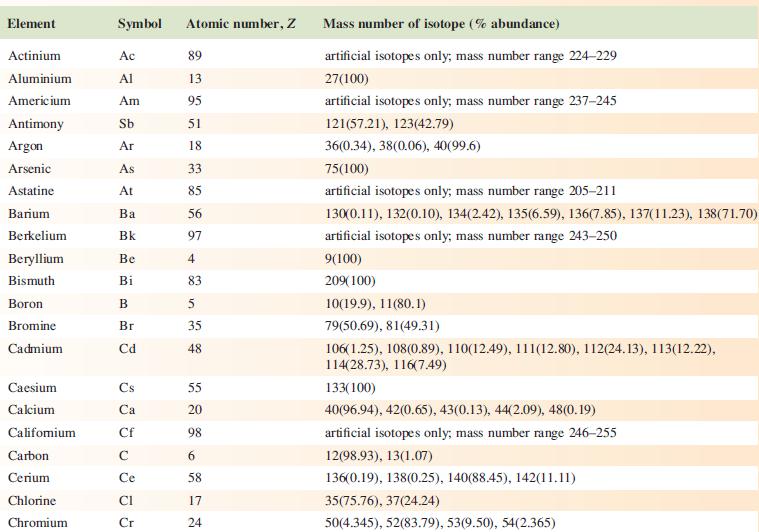
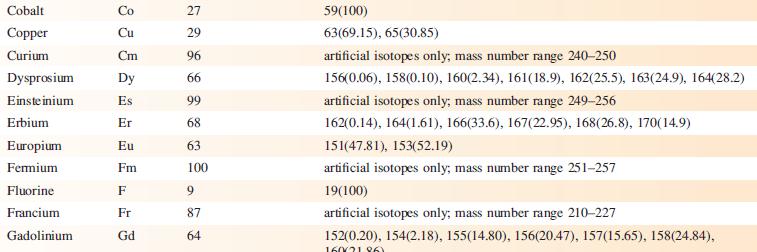
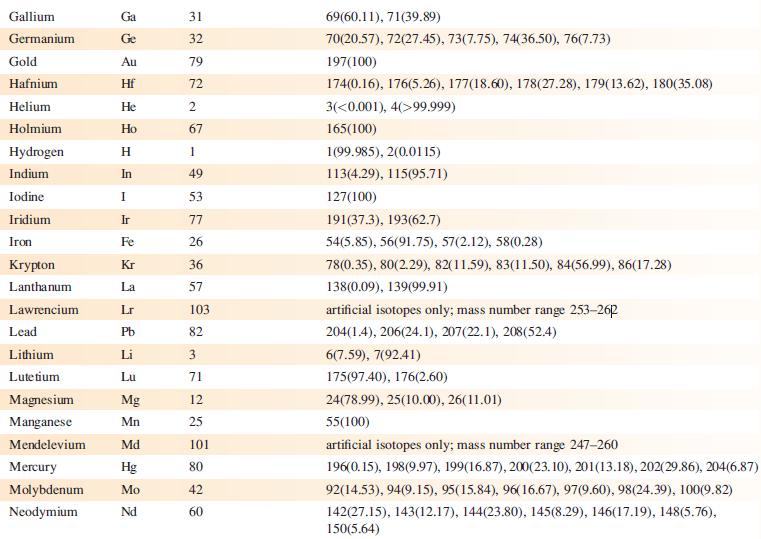
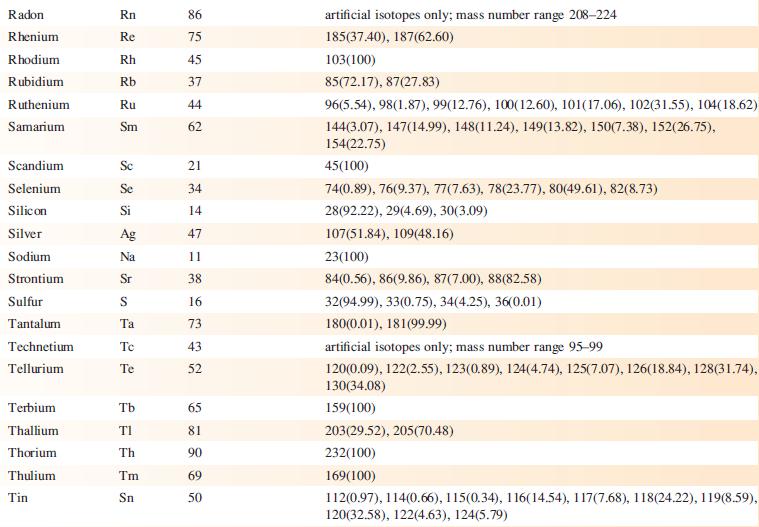
The EI mass spectrum of lead(II) acetate shows four peak envelopes, each with an isotope pattern characteristic of Pb. The most intense peak in each envelope appears at m/z 326.0, 267.0, 224.0 and 208.0, respectively.
(a) By using Appendix 5, sketch the pattern of each peak envelope.
(b) Assign the peaks.
Data from Appendix 5

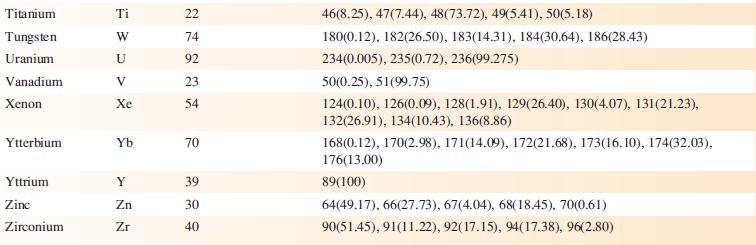
Transcribed Image Text:
Element Actinium Aluminium Americium Antimony Argon Arsenic Astatine Barium Berkelium Beryllium Bismuth Boron Bromine Cadmium Caesium Calcium Califomium Carbon Cerium Chlorine Chromium Symbol Atomic number, Z Ac Al Am Sb Ar As At Ba Bk Be Bi B Br Cd Cs Ca Cf с Ce CI Cr 89 13 95 51 18 33 85 56 97 4 83 5 35 48 55 20 98 6 58 17 24 Mass number of isotope (% abundance) artificial isotopes only; mass number range 224-229 27(100) artificial isotopes only; mass number range 237-245 121(57.21), 123(42.79) 36(0.34), 38(0.06), 40(99.6) 75(100) artificial isotopes only; mass number range 205-211 130(0.11), 132(0.10), 134(2.42), 135(6.59), 136(7.85), 137(11.23), 138(71.70)) artificial isotopes only; mass number range 243-250 9(100) 209(100) 10(19.9), 11(80.1) 79(50.69), 81(49.31) 106(1.25), 108(0.89), 110(12.49), 111(12.80), 112(24.13), 113(12.22), 114(28.73), 116(7.49) 133(100) 40(96.94), 42(0.65), 43(0.13), 44(2.09), 48(0.19) artificial isotopes only; mass number range 246-255 12(98.93), 13(1.07) 136(0.19), 138(0.25), 140(88.45), 142(11.11) 35(75.76), 37(24.24) 50(4.345), 52(83.79), 53(9.50), 54(2.365)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Here are the visual representations of the isotope patter...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rationalize the indicated fragments in the EI mass spectrum of each of the following molecules by proposing a structure of the fragment and a mechanism by which it is produced. (a)...
-
Suggest a structure for each of the ions corresponding to the following peaks in the EI mass spectrum of ethyl bromide, and give a mechanism for the formation of each ion. (The numbers in parentheses...
-
Propose reasonable fragmentation mechanisms that explain why The EI mass spectrum of benzoic acid shows major peaks at m/z = 105 and m/z = 77.
-
Two slot machines offer to double your money 3 times out of 5. Machine A takes $10 bets and Machine B takes $100 bets on each occasion. A risk-averse investor prefers to bet on A) Machine A B)...
-
Journalize the following transactions for the Pool Doctor. Explanations are not required. a. The Pool Doctor sold $63,000 of merchandise to By the River Spas, Inc., on account, terms 4/15, n/60. The...
-
A certain stock has a beta of 1.3. If the risk-free rate of return is 3.2 percent and the market risk premium is 7.5 percent, what is the expected return of the stock? What is the expected return of...
-
Using the variance data for Menounos Manufacturing Co., prepare an income statement through gross profit for the year ended December 31, 20Y6. Assume the company sold 3,000 units at $100 per unit.
-
Assume that the auditors encountered the following sepa-rate situations when deciding on the report to issue for the current- year financial statements. 1. The auditors decided that sufficient...
-
1. An object of mass of 4 kg is resting at a height of 2 m and then falls to the ground. a. Model the energy of the object-Earth system with Energy Bar Charts when at the initial height and ground...
-
Both positive and negative-ion ESI mass spectra of [Me 4 Sb][Ph 2 SbCl 4 ] were recorded. In one spectrum, peaks at m/z 181 (100%), 182 (4.5%), 183 (74.6%) and 184 (3.4%) were observed. The other...
-
TheESImass spectrum(positivemode) of the compound shown below exhibits two peaks at m/z 299.2 (base peak) and 321.1. (a) What is a base peak? (b) Suggest how the observed peaks arise. [Data: C.J....
-
Rusties Company recently implemented an activity-based costing system. At the beginning of the year, management made the following estimates of cost and activity in the company's five activity cost...
-
Assume that we know that for the period 19262010 the yield component for common stocks was 3.99 percent and that the cumulative wealth index was $2,420.46. The cumulative wealth index value for the...
-
A questionnaire was sent to 500 of a dry cleaners customers to solicit their opinions about service received. Twenty-three customers were found to be unhappy with the service. On this evidence, can...
-
You are working for a VCR manufacturer. There are three shifts in the plant: morning shift, evening shift, and midnight shift. The manager suspects that the midnight shifts productivity is lower than...
-
The dean of the school of business wants the proportion of A grades given out by his faculty members to be around 10 %. He randomly surveys 2,000 students in 50 classes and finds that of the 2,000...
-
A food company claims that its new product, low-fat yogurt, is 99 % fat-free. The management wants to keep the proportion of bad (not 99 % fat-free) products below 2 %. Inspectors check 500 cups of...
-
HighTech Inc. was a small company started by four entrepreneurs a few years ago. They each initially invested $200,000 and sold $1 million in preferred shares to a wealthy private investor. The...
-
Controls can be identified based on their function. The functions are preventive, detective, and corrective. A. True B. False
-
In their 2009 paper Icon of chemistry: The periodic system of chemical elements in the new century (Angew. Chem. Int. Ed., 2009, 48, 3404), S. Wang and W. Schwarz claim that the periodic system of...
-
According to the Born interpretation, the probability of finding an electron in a volume element dt is proportional to 2 d. (a) What is the most probable location of an electron in an H atom in its...
-
Survey the early and modern proposals for the construction of the periodic table. You should consider attempts to arrange the elements on helices and cones as well as the more practical...
-
2023 Tax Rate Schedules Individuals Schedule X-Single If taxable income is over: But not over: $ 0 $ 11,000 $ 11,000 $ 44,725 $ 44,725 $ 95,375 $ 95,375 $ 182,100 $231,250 $ 578,125 $ 182,100 $...
-
The following table lists several corporate bonds being traded in the later part of 2021. Treat these as zero coupon bonds, as in Example 2. Company Alphabet AT&T Ford Goldman Sachs Verizon Wells...
-
The detailed budgets for baseball pants is presented below. EXHIBIT 5.5 C&C Sports' sales forecast. 1 B D E F G C&C Sports H K M N 2 Sales Forecast 2021 3 4 Jan Feb Mar 5 Baseball pants 38,000 20,000...

Study smarter with the SolutionInn App


