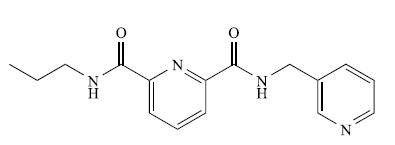
TheESImass spectrum(positivemode) of the compound shown below exhibits two peaks at m/z 299.2 (base peak) and 321.1.
Question:
TheESImass spectrum(positivemode) of the compound shown below exhibits two peaks at m/z 299.2 (base peak) and 321.1.
(a) What is a ‘base peak’?
(b) Suggest how the observed peaks arise. [Data: C.J. Sumby et al. (2009) Tetrahedron, vol. 65, p. 4681.]
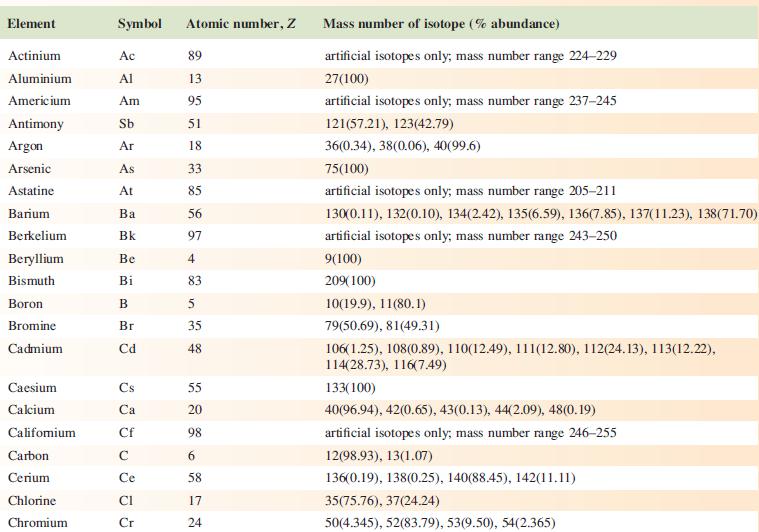
Data from Appendix 5

Transcribed Image Text:
H IZ IN Z O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
a What is a base peak In mass spectrometry the base peak is the peak with the highest intensity on the mass spectrum It is assigned a relative intensi...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In Problems 1542, solve each system of equations using Cramers Rule if it is applicable. If Cramers Rule is not applicable, write, Not applicable. x + 3 2 - + -3x + 3y - 2z 2 = -2 z = -5 5
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The compound shown below has been advertised as a dietary supplement that purportedly prevents obesity, heart disease, and the ill effects of aging. Based on its structure, what physiological...
-
The Chief Financial Officer at Ford Motor Company is said to usea hybrid-costing system. Define the hybrid-costing system. Explainthe advantages to this company to use this system. I want a 10 page 2...
-
Journalize the following transactions for the Jamin Jimmys music store. a. Purchased $8,900 of merchandise on account, terms 3/15, n/30, FOB shipping point. b. Paid $180 to the freight company for...
-
Could I Industries just paid a dividend of $1.10 per share. The dividends are expected to grow at a 20 percent rate for the next six years and then level off to a 4 percent growth rate indefinitely....
-
What would happen to the SML graph in Figure 8.8 if expected inflation increased or decreased? Figure 8.8 268 269 270 271 272 273 274 275 A Required Rate of Return TH-13.0% SML: r, RF+RPM * b D E F H...
-
The Marlin Company produces plastic bottles to customer order. The quality inspector randomly selects four bottles from the bottle machine and measures the outside diameter of the bottle neck, a...
-
The bar BC exerts a force on the cables at B that is directed along the line from C to B. (Figure 1) The mass of the suspended load is 190 kg. Suppose that = 20 and = 20. Part A What is the tension...
-
The EI mass spectrum of lead(II) acetate shows four peak envelopes, each with an isotope pattern characteristic of Pb. The most intense peak in each envelope appears at m/z 326.0, 267.0, 224.0 and...
-
The ESI mass spectrum (positive mode) of the complex shown below contained a peak envelope with m/z 527.9 (100%), 528.9 (15%), 529.9 (46%), 530.9 (7%), 531.9 (0.5%). A group of peaks of low intensity...
-
Explain why being able to obtain areas under the standard normal curve is important.
-
Assume that youre taking a part-time job in a zoo. You are called upon to inspect a new cage built to contain a ferocious lion. Do you set up the null hypothesis that the cage is safe or that the...
-
A new medicine was invented to treat hay fever, but the new drug was found to have unpleasant side effects. An experiment on 5,000 women and 4,000 men showed that 100 women and 60 men suffered side...
-
A production manager is interested in the number of defects in batches derived from different production processes. He examines a random sample drawn from each process and records the following data:...
-
Refer to question 41 to find the power of a 10 % level test when the true population mean mileage is 36 miles per gallon. Question 41 An automobile manufacturer claims that a new car gets an average...
-
You are working for a consumer rights organization. You are interested in knowing whether the milk contained in 16-oz (1-pint) bottles really weighs 16 oz. You do not want to accuse the packer of...
-
The following are selected account balances from Eastern Shore Ltds trial balance on December 31, 2016: Required: a. Prepare Eastern Shores statement of income and comprehensive income and statement...
-
a. Show that the expansion of q(x) in ascending powers of x can be approximated to 10 2x + Bx 2 + Cx 3 where B and C are constants to be found. b. Find the percentage error made in using the series...
-
What is the ratio of the energy of an electronic ground-state He + ion to that of a Be 3+ ion?
-
In the paper What can the BohrSommerfeld model show students of chemistry in the 21st century? (M. Niaz and L. Cardellini, J. Chem. Educ., 2011, 88, 240) the authors use the development of models of...
-
(a) Construct the form of each molecular orbital in linear [HHeH] 2+ using 1s basis atomic orbitals on each atom and considering successive nodal surfaces. (b) Arrange the MOs in increasing energy....
-
Two mutually exclusive projects are under consideration with the details shown. The company's 'required rate of return for projects of this risk level is 13%. using this information, answer the...
-
Refer to the Table 1. The six-month forward bid price for dollars as denominated in Japanese yen is:
-
8. Consider the following static game: 2 L R U 1 1.2 where D-1,0,0 takes two values, 0 or 2, and y takes two values, 1 or 3. Think of as the type of player 1 and of y as the type of player 2. Thus,...

Study smarter with the SolutionInn App


