The first list below contains words or phrases, each of which has a partner in the second
Question:
The first list below contains words or phrases, each of which has a ‘partner’ in the second list, e.g. ‘sodium’ in the first list can be matched with ‘metal’ in the second list. Match the ‘partners’; there is only one match for each pair of words or phrases.
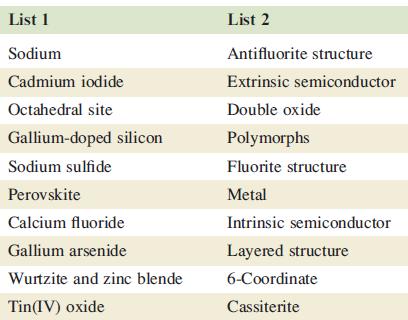
Transcribed Image Text:
List 1 Sodium Cadmium iodide Octahedral site Gallium-doped silicon Sodium sulfide Perovskite Calcium fluoride Gallium arsenide Wurtzite and zinc blende Tin(IV) oxide List 2 Antifluorite structure Extrinsic semiconductor Double oxide Polymorphs Fluorite structure Metal Intrinsic semiconductor Layered structure 6-Coordinate Cassiterite
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Lets match the partners from List 1 to List 2 Sodium Metal Cadmium iodide ...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The first list in the table on the left contains symbols or phrases, each of which has a partner in the second list. Match the partners; there is only one match for each pair of symbols or phrases.
-
The first list below contains the formula of a hydride. Each has a partner in the second list of phrases. Match the partners; there is only one match for each pair. Structural descriptions refer to...
-
Please help me with this Java Assignment. Solving the game of Boggle can be done elegantly with recursion and backtracking. Backtracking is a technique whereby an algorithm recognizes it is...
-
A car is initially travelling at 3 0 m / s , which is above the speed limit . The driver sees a speed limit trap ahead and applies the brakes for 5 seconds, causing the car to slow down by 2 m / s...
-
China has been a recipient of considerable investment recently. However, little research has been conducted by the green technology company for which you work concerning the exact nature of socially...
-
Phil Williams and Liz Johnson are 60% and 40% partners, respectively, in Williams & Johnson Partnership. Their beginning basis is $33,000 for Phil and $31,000 for Liz. The partnership had the...
-
What are the different types of consulting and litigation support activities for fraud and forensic accounting professionals?
-
Data related to the expected sales of mountain bikes and road bikes for Cycle Sports Inc, for the current year, which is typical of recent years, are as follows: Products......Unit Selling...
-
You invest $300,000 for new equipment at a manufacturing facility. The equipment is expected to produce $125,000 per year in revenue. The expenses for operating the equipment are $45,000/year. There...
-
Salts containing the [N 5 ] + and [N 3 ] ions are known (see Section 15.5), but (as of 2016) the ionic salt [N 5 ] + [N 3 ] has not been isolated. (a) Use the VBT approach to estimate the lattice...
-
Discuss the interpretation of the following: (a) f H o (298 K) becomes less negative along the series LiF, NaF, KF, RbF, CsF, but more negative along the series LiI, NaI, KI, RbI, CsI. (b) The...
-
Discuss how sustainability might be incorporated into a process or value chain improvement initiative. Try to find an example and summarize it in a manner similar to that in the box feature on...
-
How does investing more heavily in current assets (while not increasing the firms current liabilities) decrease both the firms risk and its expected return on its investment?
-
Donna, a chartered financial analyst (CFA) wants to advise her client about investment in preference shares. The client needs to invest in either Queens Plc. or Royal Plc.s preferred shares. Queens...
-
What is an LLC?
-
Explain in words the calculation of a firms weighted average cost of capital. What exactly are we averaging, and what do the weights represent?
-
Explain why large and growing firms tend to choose the corporate form.
-
Identify two key differences between the capital cost allowance (CCA) system and accounting depreciation.
-
How has the too-big-to-fail policy been limited in the FDICIA legislation? How might limiting the too-big-to-fail policy help reduce the risk of a future banking crisis?
-
Determine the position of the compound Rb 2 In 3 on a Ketelaar diagram and hence predict the type of bonding in this phase. By comparison with known structural motifs from boride cluster chemistry...
-
Indium tin oxide has important applications as a transparent conducting oxide. Which oxide(s) could be doped with Al 2 O 3 to produce a material with similar properties?
-
Indium forms a chloride of the stoichiometry In 5 Cl 9 . Rewrite this formula in terms of the oxidation states commonly adopted by indium. Describe the bonding in this compound given that it has been...
-
3. (Convergence property of power methods) Let A = () Take the initial guess 90 =[34]. Perform the power method by hand and explain why the sequence q fails to converge by calculating qk and Ak. 4....
-
3. Move 1: Rotate 270 clockwise around the origin. Move 2: Reflect over the y-axis. 4. Move 1: Translate 3 units right and 3 units up. Move 2: Reflect over the y-axis List the coordinates of the last...
-
Q-20 Solve the triangle ABC given C = 40, b = 23 cm and a = 19 cm. A-20 Q-21 A-21 97 28 cm 22 cm x

Study smarter with the SolutionInn App


