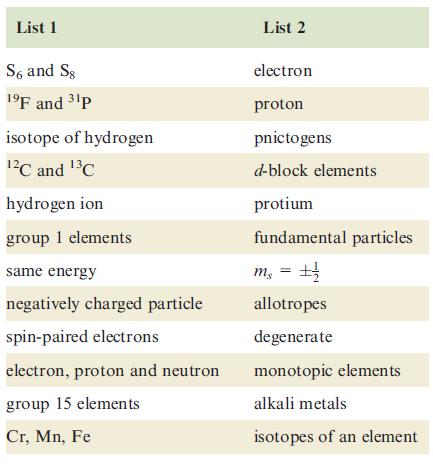
The first list in the table on the left contains symbols or phrases, each of which has
Question:
The first list in the table on the left contains symbols or phrases, each of which has a ‘partner’ in the second list. Match the ‘partners’; there is only one match for each pair of symbols or phrases.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
Symbol or phr...View the full answer

Answered By

Usman Nasir
I did Master of Commerce in year 2009 and completed ACCA (Association of Chartered Certified Accountants) in year 2013. I have 10 years of practical experience inclusive of teaching and industry. Currently i am working in a multinational company as finance manager and serving as part time teacher in a university. I have been doing tutoring via many sites. I am very strong at solving numerical / theoretical scenario-based questions.
4.60+
16+ Reviews
28+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The first list below contains words or phrases, each of which has a partner in the second list, e.g. sodium in the first list can be matched with metal in the second list. Match the partners; there...
-
Create a chart of accounts for Raymond Autobody Shop using the standard numbering system. Each account is separated by a factor of 10. For example, the first asset account will be 100 and the next...
-
In Data 2.1 on page 46, we introduce a study in which people were asked whether they agreed or disagreed with the statement that there is only one true love for each person. Table 7.27 gives a...
-
Tin - Can, Inc. Aircraft ( TCAI ) R&D Project Management Problem Your group is hired to help TCAI Project Manager to solve the following problem. Using the activity time estimates and activity...
-
The transactions for Grinko, Inc., for the month of June 2012 have been posted to the accounts as follows: Requirements 1. Prepare the journal entries that served as the sources for the six...
-
Why are most labor unionswhose constituents receive wages substantially above the minimum wagestrong supporters of the minimum wage? Why might unions composed of skilled workers who are pure...
-
The plaintiffs, Lee and Yoon, were the parents of H.L., a South Korean citizen who attended high school in Idaho as part of an exchange program. With permission from both plaintiffs and his host...
-
Modular houses are Homes, Inc.s specialty. The companys best-selling model is a three-bedroom, 1,400-square-foot house with an impressive front entrance. Last year, the standard costs for the six...
-
A violet ray of light leaves a medium with refractive index of 6.46 at an angle of 0.07 radians to the normal and enters a second medium with refractive index of 22.04. At what angle to the normal...
-
Suggest explanations for the following. (a) High values of ionization energies are associated with the noble gases. (b) The enthalpy changes associated with the attachment of the first and second...
-
What is the evidence that the aufbau principle is only approximately true?
-
What is vector analysis?
-
What are the two types of asset-price bubbles? Which type poses a bigger threat to the financial system? Why?
-
What is the foreign exchange market? Describe the two types of transactions that take place in this market.
-
How do fixed, floating, and managed (dirty) float exchange rate regimes differ?
-
Financial regulators have been working to improve transparency and reduce risk in the derivatives market. How do you think increased transparency will affect financial intermediaries that trade...
-
What modifications to the intertemporal choice theory have been suggested by the random walk hypothesis and behavioral economics?
-
If bonds were issued at a discount, explain how the amount of cash received on issuance would compare with the face value of the bonds.
-
Represent each of the following combination of units in the correct SI form using an appropriate prefix: (a) m/ms, (b) k m, (c) k s /mg, and (d) k m N.
-
Superacids are well established but superbases also exist and are usually based on hydrides of Group 1 and Group 2 elements. Write an account of the chemistry of superbases.
-
Calculate the equilibrium concentration of H 3 O + in a 0.10 M solution of butanoic acid (K a = 1.86 10 5 ). What is the pH of this solution?
-
In their paper The strengths of the hydrohalic acids (J. Chem. Educ., 2001, 78, 116), R. Schmid and A. Miah discuss the validity of literature values of the pK a values for HF, HCl, HBr, and HI. (a)...
-
Find f(g(x)) and g(f(x)) and determine whether the pair of functions f and g are inverses of each other. f(x)=/x-6 and g(x)=x5+6 f(g(x)) =
-
Given the following functions, find and simplify (f + g)(1). f(x) = -2x22x Do not include "(f+g)(1) = " in your answer. Provide your answer below: g(x) = x - 2
-
f(x+h) - f(x) Find the difference quotient of f(x) = x-7; that is find , h0. Be sure to simplify. h

Study smarter with the SolutionInn App


