Use Fig. 6.12 to find the approximate potential of an aerated lake at pH = 6. With
Question:
Use Fig. 6.12 to find the approximate potential of an aerated lake at pH = 6. With this information and Latimer diagrams from Resource section 3, predict the species at equilibrium for the elements
(a) Iron,
(b) Manganese,
(c) Sulfur.
Figure 6.12.
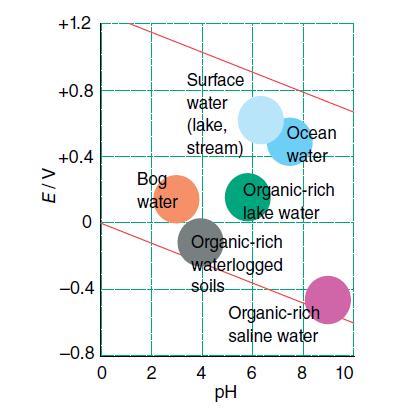
Transcribed Image Text:
E/V +1.2 +0.8 +0.4 0 -0.4 -0.8 Bog water Surface water (lake, stream) 02 4 Organic-rich waterlogged soils S pH Organic-rich lake water Ocean water Organic-rich saline water 8 6 10
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
The approximate potential of an aerated lake at pH 6 is around 02 V a For iron the most stable oxidation state at this potential is Fe2 This is becaus...View the full answer

Answered By

User l_998468
I have extensive tutoring experience, having worked as a private tutor for over three years. I have tutored students from different academic levels, including high school, undergraduate, and graduate levels. My tutoring experience has taught me to be patient, attentive to student needs, and effective in communicating difficult concepts in simple terms.
I have a strong background in statistics, probability theory, data analysis, and data visualization. I am proficient in using statistical software such as R, Python, and SPSS, which are commonly used in academic research and data analysis. Additionally, I have excellent communication and interpersonal skills, which enable me to establish rapport with students, understand their learning styles, and adapt my teaching approach to meet their needs.
I am passionate about teaching and helping students achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Use Fig. 17.12 to find the approximate coefficient of volume expansion of water at 2.0C and at 8.0C.
-
Use Fig. 17.12 to find the approximate coefficient of volume expansion of water at 2.0C and at 8.0C.
-
The Bell Curve (Free Press, 1994), written by Richard Herrnstein and Charles Murray (H&M), is a controversial book about race, genes, IQ, and economic mobility. The book heavily employs statistics...
-
Determine the force in member GC of the truss and state if this member is in tension or compression. Units Used: kip = 103 lb Given: F1 = 1000 lb F2 = 1000 lb F3 = 1000 lb F4 = 1000 lb a = 10 ft =...
-
Due to the H1N1 flu outbreak, the demand for hand sanitizer has tripled. Should Johnson &Johnson increase production of their Purell hand sanitizer? Should it invest in doubling production capacity?
-
1. What types of businesses would prosper in China? Why? 2. What are the challenges and risks associated with doing business in China? 3. What steps should Shippertake to address these challenges and...
-
Web-based exercise. If you did any of Exercises 13.21 to 13.30, check your calculations using the Normal Curve applet described in the previous exercise.
-
Notes Payable On December 1, 2007 Insto Photo Company purchased merchandise, invoice price $25,000, and issued a 12%, 120-day note to Ringo Chemicals Company. Insto uses the calendar year as its...
-
AutoSave CH 2 ExcelTemplateAssignment CO2 (1) - Protected View Excel Search File Home Insert Page Layout Formulas Data Review View Help DYMO Label QuickBooks O PROTECTED VIEW Be careful--files from...
-
Draw a Frost diagram for mercury in acid solution, given the following Latimer diagram: Comment on the tendency of any of the species to act as an oxidizing agent, a reducing agent, or to undergo...
-
Using the following Latimer diagram, which shows the standard potentials for sulfur species in acid solution (pH = 0), construct a Frost diagram and calculate the standard potential for the HSO 4 /S...
-
Mary Ann Singleton was the librarian at a maximum security prison located in Tazewell County, Virginia. About four times a week, Gene Shinault, assistant warden for operations, insistently...
-
The pulse rates of 152 randomly selected adult males vary from a low of 37 bpm to a high of 117 bpm. Find the minimum sample size required to estimate the mean pulse rate of adult males. Assume that...
-
A rod 12.0 cm long is uniformly charged and has a total charge of -23.0 C. Determine the magnitude and direction of the electric field along the axis of the rod at a point 32.0 cm from its center....
-
Hello need help with this problem. The transactions relating to the formation of Blue Company Stores Incorporated, and its first month of operations follow. a. The firm was organized and the...
-
At the beginning of the year, the net assets of Shannon Company were $492,600. The only transactions affecting stockholders equity during the year were net income of $70,200 and dividends of $15,400....
-
The claim is that smokers have a mean cotinine level greater than the level of 2.84 ng/mL found for nonsmokers. (Cotinine is used as a biomarker for exposure to nicotine.) The sample size is n = 739...
-
Refer to the International Conference on Social Robotics (Vol. 6414, 2010) study of the trend in the design of social robots, Exercise 3.10. Recall that in a random sample of 106 social robots, 63...
-
The registrar of a college with a population of N = 4,000 full-time students is asked by the president to conduct a survey to measure satisfaction with the quality of life on campus. The following...
-
Give a summary of how conventional superconductors have impacted on our lives. Include both scientific and more general applications.
-
Legislation requires the phasing out of incandescent lighting. How does inorganic chemistry contribute to the development of solid state lighting?
-
(a) Describe the structure of lithium nitride and explain how it is able to function as a lithium ion conductor. The structures of Li 3 P and Li 3 As are analogous to that of the nitride. How do you...
-
Your company produces a health magazine. Its sales data for 1 - year subscriptions are as follows: Year of Operation Subscriptions Sold % Expired at Year End 2 0 2 0 $ 3 0 0 , 0 0 0 5 2 0 2 1 $ 6 4 7...
-
Problem 3 - 2 0 ( Static ) Calculate profitability and liquidity measures LO 3 - 3 , 3 - 4 , 3 - 6 Presented here are the comparative balance sheets of Hames Incorporated at December 3 1 , 2 0 2 3...
-
3 Required information [The following information applies to the questions displayed below) John and Sandy Ferguson got married eight years ago and have a seven-year-old daughter. Samantha. In 2020,...

Study smarter with the SolutionInn App


