Using the following Latimer diagram, which shows the standard potentials for sulfur species in acid solution (pH
Question:
Using the following Latimer diagram, which shows the standard potentials for sulfur species in acid solution (pH = 0), construct a Frost diagram and calculate the standard potential for the HSO4−/S8(s) couple.
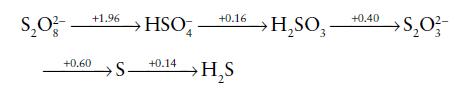
Transcribed Image Text:
$₂0²- +0.60 +1.96 +0.16 HSO +0.14 S- →H₂S → H₂SO3 +0.40 S₂0 ²/3- >
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
To construct a Frost diagram well plot the standard reduction potentials E for each species against ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The following Latimer diagrams show the standard reduction potentials E /V for some oxidation states of iron in acid and alkaline solution: (a) Plot a Frost diagram showing the states of Fe under...
-
The Latimer diagram for vanadium species in acidic (pH = 0) solution is: Using these data: (a) Calculate the potential for the reduction of VO 2+ (aq) to V(s) and write a balanced chemical equation...
-
From the following Latimer diagram (which does not correspond to standard conditions), calculate the value of E for the reaction 2HO 2 (aq) O 2 (g) + H 2 O 2 (aq). Comment on the thermodynamic...
-
Certain merchandising transactions for Yule Park Gifts are listed below. November 2: Purchased 110 souvenir calendars at $9 each from Aloha Co. for cash Nov 4: Paid shipping costs $38 for the...
-
Multiple-Choice Questions 1. Changes in prices of a good causes a. Movement along the demand curve. b. Movement along the supply curve. c. No effect to either curve. d. Both a and b 2. If the market...
-
In a recent season, hockey player Evgeni Malkin scored 27 goals in 57 games he played. Assume that his goal production stayed at that level for the next season. Find the probability that he would get...
-
What happens if you specify an invalid format string?
-
At week 24 of a project to shoot a television commercial, what should the expenditures be? If the earned value is right on schedule but the actual expenses are $9,000, what are the cost and schedule...
-
Put the argument from the following passage into standard form and construct an argument map diagram. There is no need to include unstated premises (assumptions) in your answer. Australia should not...
-
Use Fig. 6.12 to find the approximate potential of an aerated lake at pH = 6. With this information and Latimer diagrams from Resource section 3, predict the species at equilibrium for the elements...
-
Calculate the equilibrium constant of the reaction from the standard potentials Aut(aq) + 2 CN- (aq) [Au(CN)](aq)
-
How are permissions used to control access to data?
-
PunchTab Inc - venture capital (MDV) vs. Angel Investor. Which is the best investment?
-
Conditions: Imagine that you just started working for a financial organization that provides several financial calculators as web services that are available to third-party developers. Kelsey has...
-
Describing the relationships between the various financial statements. How does one transfer information to the other statements? What stories do they tell?
-
Define the two possible types of capital rationing, and discuss how capital rationing affects the attainment of management's goal of maximizing shareholders' value.
-
In which type of hedge fund investment, managers buy or sell securities and then hedge part or all of the associated risks? Question 7Answer Distressed securities Convertible arbitrage strategies...
-
Calculate E cell for the following half reaction: AgI(s) + e - Ag(s) + I 2 Reference the Ksp value for AgI and the standard reduction potential for Ag+.
-
The cost curve for the city water supply is C(Q) = 16 + 1/4 Q2, where Q is the amount of water supplied and C(Q) is the cost of providing Q acre-feet of water. (An acre-foot is the amount of water...
-
(a) Explain how single-walled carbon nanotubes are classified in zigzag and armchair tubes. (b) What inherent properties of single-walled carbon nanotubes make those formed by arc discharge or laser...
-
The properties of graphene nanoribbons depend upon their edge structure. What are the different edge structures and how do they arise?
-
Graphene is insoluble in common solvents. Describe ways in which the material can be solubilized.
-
Suppose you sell a fixed asset for $10,000 when its book value is $2,000. If your company's marginal tax rate is 35 percent, what will be the effect on cash flows of this sale (i.e., what will be the...
-
Calculate the E( R i ) & ( R i ) 2. Show in the table below and with a graph and equation the return-risk combinations that can be obtained with a risky stock portfolio with E(R) = 20% and (Rp) = 8%...
-
https://hbr.org/2019/01/case-study-when-two-leaders-on-the-senior-team-hate-each-other Please answer all questions below after reading the case study 1. Explain a negotiation strategy to resolve the...

Study smarter with the SolutionInn App


