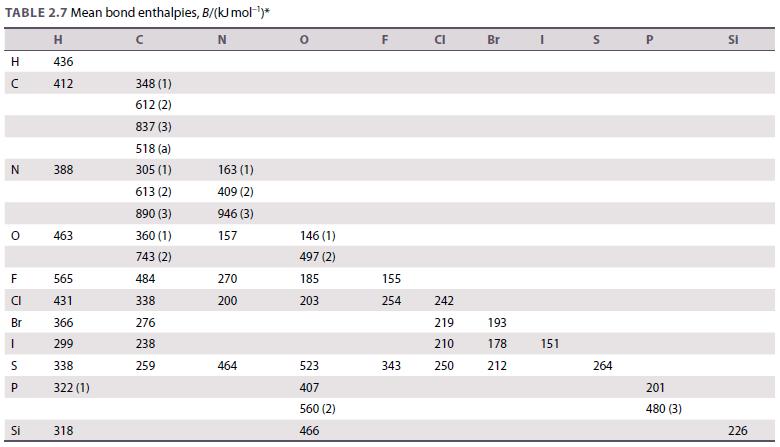
Use the data in Table 2.7 to calculate the standard enthalpy of the reaction 2 H 2
Question:
Use the data in Table 2.7 to calculate the standard enthalpy of the reaction 2 H2(g) + O2(g) → 2 H2O(g). The experimental value is −484 kJ. Account for the difference between the estimated and experimental values.
Table 2.7.
Transcribed Image Text:
TABLE 2.7 Mean bond enthalpies, B/(kJ mol-¹)* H N 436 412 H с N O F CI Br LL 1 S P Si 388 463 565 431 366 299 338 322 (1) 318 348 (1) 612 (2) 837 (3) 518 (a) 305 (1) 613 (2) 890 (3) 360 (1) 743 (2) 484 338 276 238 259 163 (1) 409 (2) 946 (3) 157 270 200 464 146 (1) 497 (2) 185 203 523 407 560 (2) 466 F 155 254 343 CI 242 219 210 250 Br 193 178 212 I 151 S 264 P 201 480 (3) SI 226
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To calculate the standard enthalpy of the reaction 2 H2g O2g 2 H2Og using the data in Table 27 we will use Hesss Law which states that the overall ent...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
In the following exercises, you will use the data in the Solmaris Condominium Group database shown in Figures 1-21 through 1-25. (If you use a computer to complete these exercises, use a copy of the...
-
The personnel director of a firm has developed two tests to help determine whether potential employees would perform successfully in a particular position. To help estimate the usefulness of the...
-
The combustion of 0.4196 g of a hydrocarbon releases 17.55 kJ of heat. The masses of the products are CO2 = 1.419 g and H2O = 0.290 g. (a) What is the empirical formula of the compound? (b) If the...
-
9:49 X Bank Reconciliation Assignment 1 PDF 150 KB Assignment #1 - Bank Reconciliation This assignment has 33 total marks. This assignment is worth 5% of your final grade. This assignment is due...
-
Bond P is a premium bond with a 10 percent coupon. Bond D is a 4 percent coupon bond currently selling at a discount. Both bonds make annual payments, have a YTM of 7 percent, and have eight years to...
-
(a) Find a linear function that contains the points (-2, 3) and (1, -6). What is the slope? What are the intercepts of the function? Graph the function. Be sure to label the intercepts. (b) Find a...
-
Mrs. Palsgraf was waiting for a train on a platform of a railroad. When a different train came into the station, two men ran to get on that train before it left the station. While one of the men...
-
Sandy Shores Time Shares is one of the largest time-sharing and rental brokers for vacation cottages along the North Carolina coast. After 10 successful years of matching up owners and renters, Sandy...
-
Anika who lives in Vancouver, purchased a piece of equipment from Australia for A$45,400. He was charged 19% duty, 5% GST, and 7% PST to import it. Calculate the total cost of the equipment. Assume...
-
Use the Ketelaar triangle in Fig. 2.28 and the electronegativity values in Table 1.7 to predict what type of bonding is likely to dominate in (a) BCl 3 , (b) KCl, (c) BeO. Figure 2.28. Table 1.7. 3 2...
-
Use the covalent radii in Table 2.6 to calculate the bond lengths in (a) CCl 4 (177 pm), (b) SiCl 4 (201 pm), (c) GeCl 4 (210 pm). (The values in parentheses are experimental bond lengths and are...
-
Option-adjusted duration and effective duration are alternative measures used by analysts to evaluate fixed-income securities with embedded options. Briefly describe each measure, and how to apply...
-
Consider the ultimatum game: The proposer offers a split of $10 between himself and the respondent. The proposer can choose any split (x, 10 x), where x [0, 10] is the amount the respondent gets...
-
Why do so many U.S. firms build their facilities in other countries in their supply chain designs?
-
(a) (b) Draw the block diagram representing the Harvard computer architecture, showing proper labels of different blocks and bus interconnections. [5 Marks] Most PIC18 MCUs provide data memory of up...
-
Our client, Vicki Farthing, has decided to purchase, personally, a used building in 2023 to be used in her new proprietorship business, which is considered to have started with preliminary work on...
-
You decide to deposit $171 monthly in a 2.9% annual interest bearing account over the next 15 years. How much will have accumulated in the account at the end of the period?
-
A real estate agent looks over the 15 listings she has in a particular zip code in California and finds that 80% of them have swimming pools. a) Check the assumptions and conditions for inference on...
-
The unadjusted trial balance of Secretarial Services is as follows: SECRETARIAL SERVICES Unadjusted Trial Balance as at 31 December 2017 Account Debit Credit Cash at bank Office supplies Prepaid...
-
The industrial manufacture of NH 3 from N 2 and H 2 is carried out on a huge scale using heterogeneous catalysis, i.e. the reaction between gaseous N 2 and H 2 is carried out over a solid catalyst....
-
Provide explanations for the following observations. (a) In moist air, corrosion of iron is spontaneous. However, under anaerobic (O 2 free), wet conditions, corrosion of iron is only marginally...
-
A plumber directly connects a galvanized steel pipe to a copper pipe in a system that carries running water. Suggest what will happen over a period of time.
-
Problem1: Design two different braking systems with torque capacity 4kN.m. The design models should consider minimizing the total volume of the brake as possible. A conclusion should be included to...
-
5. (20%) A 3, 100 KVA, 460 V, 60 Hz,6 poles wound-rotor induction motor has the following parameters per phase: R1 0.2 ohms, R'= 0.15 ohms, X1-X2'= 0.5 ohms, Xm= 20 ohms (a) Derive the Thevenin...
-
2) Two co-axial shafts transmit 40 kW of power at a rotational speed of 1500 revolutions per minute. Coupling and decoupling of the shafts is done by a clutch with friction pads of inner and outer...

Study smarter with the SolutionInn App


