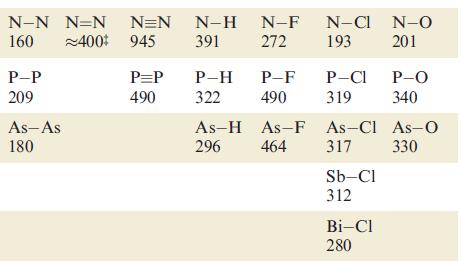
Using bond enthalpy terms from Tables 14.2 and 15.3, estimate values of r H for the
Question:
Using bond enthalpy terms from Tables 14.2 and 15.3, estimate values of ΔrHº for the following reactions
(a) 2N2 → N4 (tetrahedral structure);
(b) 2P2 → P4 (tetrahedral structure);
(c) 2C2H2 → C4H4 (tetrahedrane, with a tetrahedral C4 core).
Table 14.2.
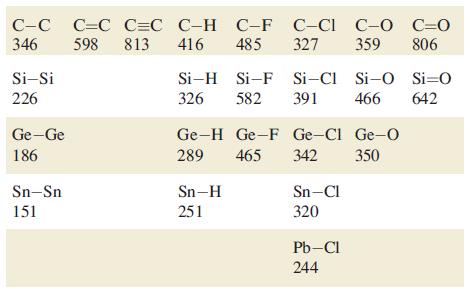
Table 15.3.
Transcribed Image Text:
C-C_C=CC=C_C-H C-F C-CI C-0_C=0 598 813 416 485 327 359 806 346 Si-Si 226 Ge-Ge 186 Sn-Sn 151 Si-H Si-F Si-Cl Si-O Si=0 582 391 466 326 642 Ge-H Ge-F 289 465 342 350 Ge-Cl Ge-0 Sn-H 251 Sn-Cl 320 Pb-Cl 244
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
To estimate the values of rH standard enthalpy change for the given reactions using bond enthalpy te...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using data from Appendix 4, calculate ÎHo, ÎSo, and DGo for the following reactions that produce acetic acid: Which reaction would you choose as a commercial method for producing acetic...
-
For the following reactions at constant pressure, predict if H . E, H , E, or H = E. a. 2HF(g) H2(g) + F2(g) b. N2(g) + 3H2(g) 2NH3(g) c. 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(g)
-
Using the data in Appendix 3, calculate the standard entropy changes for the following reactions at 25C: (a) S(s) + O2(g) SO2(g) (b) MgCO3(s) MgO(s) + CO2(g)
-
Consider each of the following situations. Indicate whether (and why or why not) you think that the governement should account for the transactions and resources in an agency fund, a governmental...
-
A number of business transactions carried out by Smalling Manufacturing Company are as follows: a. Borrowed money from a bank. b. Sold land for cash at a price equal to its cost. c. Paid a liability....
-
Find the result of each operation on the given point (represented by Cartesian coordinates): (a) C 2(z) i h (1,1,1). (b) S 2(y) h (1,1,0).
-
On April 23, 2014, Calvin Loyer admitted his wife, Edeltrud Loyer, to a nursing home administered by Signature Healthcare. During the admissions process, Calvin signed an arbitration agreement...
-
The mean preparation fee H&R Block charged retail customers last year was $183 (The Wall Street Journal, March 7, 2012). Use this price as the population mean and assume the population standard...
-
Country A is endowed with 18 units of good x and 1 unit of good y, while country B is endowed with 2 units of good x and 7 units of good y. Both countries consume x and y in fixed and equal...
-
Lead-acid batteries accounted for 69% of all lead consumed in the US in 2015. (a) Complete the cell reaction given below (not balanced on the left-hand side) and show that the oxidation state changes...
-
The glass industry manufactures millions of tonnes of glass per year. (a) Only certain element oxides form glasses. Explain why this is, giving examples of what are termed in the glass industry as...
-
Why is the American economy called a mixed economy?
-
At the beginning of the year, a company estimates total direct materials costs of $1,050,000 and total overhead costs of $1,200,000. If the company uses direct materials costs as its activity base to...
-
During January, its first month of operations, Sheridan Company accumulated the following manufacturing costs: raw materials purchased $5,200 on account, factory labor incurred $6,600, and factory...
-
Environmental Accounting and Sustainability Reporting - Brief Information Step 1: Data Collection and Measurement Environmental accounting involves the systematic collection and measurement of data...
-
Accounting for Contingencies and Provisions In accounting, contingencies and provisions play a crucial role in preparing financial statements and ensuring accurate representation of a company's...
-
When does a partnership terminate for federal tax purposes? When at least 50% of total partnership interests are sold within a 12-month period. When all operations are completely discontinued. When...
-
(1) Analyze the studentized deleted residuals in the page margin for Options 1 and 2 (see SDR1 and SDR2). (2) Is hospital 14 an outlier with respect to its y value when using Option 2? (3) Consider a...
-
The National Collegiate Athletic Association (NCAA) and the National Federation of State High School Associations (NFHS) set a new standard for non-wood baseball bats. Their goal was to ensure that...
-
Draw the B 12 unit that is a common motif of boron structures; take a viewpoint along a C 2 axis.
-
Give balanced chemical equations for the synthesis of 1,2-B 10 C 2 H 10 (Si(CH 3 ) 3 ) 2 starting with decaborane(14) and other reagents of your choice.
-
Give the IUPAC names of (a) B 10 H 14 , (b) [B 12 H 12 ] 2 , (c) Arachno-[B 12 H 14 ] 2 .
-
By defining development to include more than just growth in per capita income, it is implicitly assumed that growth in per capita income alone is not sufficient to ensure poverty reduction and...
-
Pat operated a mail order business during the year. Revenues from the business were $34,000 and the cost of merchandise purchased was 18,000 of which $4,000 was still held as inventory at December...
-
American and Mexican workers can each produce 10 T-shirts per day.An American worker can produce 18 dozen brownies per day, while a Mexican worker can produce 10 dozen brownies per day.To simplify...

Study smarter with the SolutionInn App


