(a) Derive and in terms of measurable properties. (b) dH = dU + d(PV) from the definition...
Question:
(a) Derive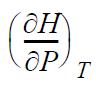
and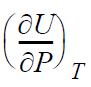
in terms of measurable properties.
(b) dH = dU + d(PV) from the definition of H. Apply the expansion rule to show the difference between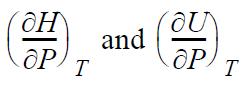
is the same as the result from part (a).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Introductory Chemical Engineering Thermodynamics
ISBN: 9780136068549
2nd Edition
Authors: J. Elliott, Carl Lira
Question Posted:





