Determine the difference C P - C V for the following liquids using the data provided near
Question:
Determine the difference CP - CV for the following liquids using the data provided near 20°C.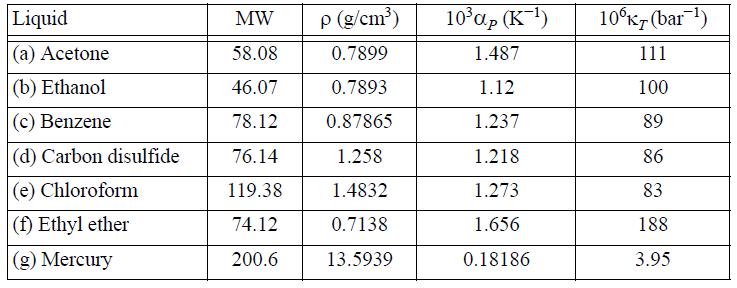

Transcribed Image Text:
Liquid (a) Acetone (b) Ethanol (c) Benzene (d) Carbon disulfide (e) Chloroform (f) Ethyl ether (g) Mercury MW 58.08 46.07 78.12 76.14 119.38 74.12 200.6 p (g/cm³) 0.7899 0.7893 0.87865 1.258 1.4832 0.7138 13.5939 10³αp (K-¹) 1.487 1.12 1.237 1.218 1.273 1.656 0.18186 10°KT (bar ¹) 111 100 89 86 83 188. 3.95
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
Liquid Molar Mass gmol Density gcm 103 K 106 bar CP CV ...View the full answer

Answered By

Marcus Solomon
I am committed to ensuring that my services always meet the clients' expectations.
4.60+
82+ Reviews
117+ Question Solved
Related Book For 

Introductory Chemical Engineering Thermodynamics
ISBN: 9780136068549
2nd Edition
Authors: J. Elliott, Carl Lira
Question Posted:
Students also viewed these Engineering questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Using the PC state daemon, calculate from first principle (a) cp (b) cv of steam at 100 kPa and 320oC. Use a 1oC difference in setting up the neighboring states. (c) What is the ratio of (cp - cv)/R...
-
prepare a post-closing trial balance. Cash Account Titles Accounts Receivable Supplies Accounts Payable Unearned Service Revenue Owner's Capital Owner's Drawings Service Revenue Salaries and Wages...
-
In what way does the patent system help society solve an externality problem?
-
Identify the direction of increase in z in each of the following cases: (a) Maximize z = x1 - x2 (b) Maximize z = - 5x1 - 6x2. (c) Maximize z = - x1 + 2x2. (d) Maximize z = - 3x1 + x2.
-
What lump-sum amount of interest will be paid on a \($10,000\) loan that was made on 1 June 2011 and repaid on 1 September 2015, with ordinary simple interest at 10% per year?
-
1. Which of the following is not subject to self-employment tax? a. Net earnings of the owner of a shoe store b. Net earnings of a self-employed lawyer c. Distributive share of earnings of a...
-
PATA PLC acquired 7 5 % of the equity interest in POTEA PLC at 1 January 2 0 1 9 for KShs . 5 0 million. The Statement of Financial Position as at 3 1 March 2 0 2 0 were as follows: PATA PLC POTEA...
-
The fundamental internal energy relation for a rubber band is dU = TdS - FdL where F is the system force, which is negative when the rubber band is in tension. The applied force is given by F applied...
-
(a) Prove (b) For an ideal gas along an adiabat, (P/P i ) = (T/ T i ) C P /R . Demonstrate that this equation is consistent with the expression from part (a). JP OT S || P TVOp
-
A neutron collides elastically with an initially stationary deuteron. Find the fraction of the kinetic energy lost by the neutron (a) In a head-on collision; (b) In scattering at right angles.
-
Describe the difficulty caused by the strict covenant to pay rent as it relates to frustration in contract law.
-
When a director enters into a contract with his or her own corporation, what precautions should be taken to ensure the validity of the contract?
-
What are the advantages and disadvantages of partnerships as opposed to sole proprietorships?
-
What is meant by intercepting a corporate opportunity?
-
Should a leasehold interest be shown as an asset (with an equal and offsetting liability) on the balance sheet of a business? If so, at what amount?
-
Compare future consumption to current consumption. Is one necessarily better than the other? Discuss.
-
Four GWU students have been selected to taste food sold by 3 different food trucks labeled as food truck A, B and C on H & 22nd Streets every Monday for 3-weeks. For each student, food trucks are...
-
From the following equilibrium constants, calculate the equilibrium constant for the reaction HO,CCO,H 2H* + C,0.
-
Assuming complete dissociation of the salts, calculate the ionic strength of (a) 0.2 mM KNO 3 ; (b) 0.2 mM Cs 2 CrO 4 ; (c) 0.2 mM MgCl 2 plus 0.3 mM AlCl 3 .
-
Find the activity (not the activity coefficient) of the (C 3 H 7 )4N + (tetrapropylammonium) ion in a solution containing 0.005 0 M (C 3 H7) 4 N + Br - plus 0.005 0 M (CH 3 )4N + Cl - .
-
Molson Coors Beverage Company issued a coupon bond 3.00 years ago. Southern Company issued a coupon bond 4.50 years ago. Both bonds pay coupons annually and mature in 9 years. The Molson Coors...
-
FastJet Airlines Corp. is considering the cash purchase of four new airliners for $20,000,000 each. These airplanes will save the company a total of $300,000 per year in operational expenses compared...
-
Review the paralegalassociations (NFPA, NALS, NALA via the internet). Answer the following questions: What are the requirements to join (how can you become a member)? Do you possess any of the...

Study smarter with the SolutionInn App


