Ethyl acetate is to be produced by a liquid phase reaction. (a) Use the shortcut vant Hoff
Question:
Ethyl acetate is to be produced by a liquid phase reaction.
(a) Use the shortcut van’t Hoff equation to calculate the expected conversion of HOAc for equimolar feeds of EtOH and HOAc in a batch reactor at 80°C.
(b) Repeat part (a) with a 3:1 ratio of EtOH to HOAc at 80°C.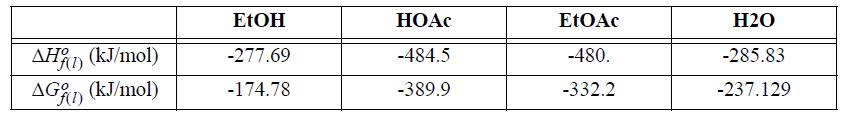
Transcribed Image Text:
AH(1) (kJ/mol) AG(1) (kJ/mol) EtOH -277.69 -174.78 HOAc -484.5 -389.9 EtOAc -480. -332.2 H2O -285.83 -237.129
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Using the vant Hoff equation we can determine the predicted conversion of acetic acid HOAc for the s...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemical Engineering Thermodynamics
ISBN: 9780136068549
2nd Edition
Authors: J. Elliott, Carl Lira
Question Posted:
Students also viewed these Engineering questions
-
A gas turbine is operated with gases (Cp = 0.992 kJ/kg K and = 1.29) entering it at 10 bar and 1025C, and leaving it at 1 bar and 550C. Assuming adiabatic flow through the turbine, calculate the...
-
For the mandrel needed in Problem 7.90, plot the sheet-metal thickness as a function of radius if the part is to be produced by shear spinning. Comment on whether or not this operation is feasible....
-
SQL, Structured Query Language, is a language used by software developers, data scientists, and anyone who works with data. There are various database management systems (DBMS) that support SQL, but...
-
What is the first question to ask when redefining quality goals? What is the purpose of what I am trying to accomplish? What is the right purpose for me to be working on? What quality goals should I...
-
1. Would you describe Starbucks production/operations technology in its retail stores as unit, mass, or process? Explain your choice. How does its production/operations technology approach affect the...
-
Air enters a diffuser with a velocity of 180 m/s and a inlet temperature of 303 K. Determine (a) The velocity of sound and (b) The flow Mach number at the diffuser inlet.
-
1. Describe "intangible" assets. 2. Is it possible for working capital to be negative? Retained earnings? Why? 3. The liquidity of assets is important to creditors. Does the liquidity of assets...
-
Are New Belgiums social initiatives indicative of strategic philanthropy? Why or why not? Although most of the companies frequently cited as examples of ethical and socially responsible firms are...
-
Solve the inequality involving absolute value. 2-2+613 Enter the exact answer in interval notation. To enter oo, type infinity. To enter U, type U. Show your work and explain, in your own words, how...
-
Two-tenths of a gram of CaCO 3 (s) is placed in a 100cm 3 pressure vessel. The vessel is evacuated of all vapor at 298 K and sealed. The reaction CaCO 3(s) = CaO (s) + CO 2(g) occurs as the...
-
Limestone (CaCO 3 ) decomposes upon heating to yield quicklime (CaO) and carbon dioxide. At what temperature does limestone exert a decomposition pressure of 1 bar?
-
A 732-kg car stopped at an intersection is rear-ended by a 1720-kg truck moving with a speed of 15.5 m/s. If the car was in neutral and its brakes were off, so that the collision is approximately...
-
Sophie Chan owns 100,000 shares of PAT Company. PAT is selling for 40 per share, so her investment is worth 4,000,000. Chan reinvests the gross amount of all dividends received to purchase additional...
-
The bond equivalent yield for a 182-day U.S. Treasury bill that has a price of \($9\),725 per \($10\),000 face value is closest to: A. 5.44%. B. 5.53%. C. 5.67%.
-
Using vertical common-size analysis and restating the balance sheets using total assets as the benchmark to analyze changes at Tab between fiscal year 2003 and fiscal year 2005, an analyst would...
-
Beta Corporation is a manufacturer of inflatable furniture. Which of the following scenarios best reflects a stable dividend policy for Beta? A. Maintaining a constant dividend payout ratio of 4050%....
-
Analysts label stock markets bubbles when market prices appear to lose contact with intrinsic values. To many analysts, the run-up in the prices of internet stocks in the US market in the 19982000...
-
Ben Fun, Inc., manufactures video games. Market saturation and technological innovations have caused pricing pressures, which have resulted in declining profits. To stem the slide in profits until...
-
What is the amount of total interest dollars earned on a $5,000 deposit earning 6% for 20 years?
-
Iminodiacetic acid forms 2:1 complexes with many metal ions: A 25.0 mL solution containing 0.120 M iminodiacetic acid buffered to pH 7.00 was titrated with 25.0 mL of 0.050 0 M Cu 2+ .Given that x2...
-
What is the chelate effect?
-
Auxiliary complexing agent. Use the equation derived in Problem 11-19. (a) Prepare a spreadsheet to reproduce the 20-, 50-, and 60-mL points in the EDTA titration of Zn 2+ in the presence of NH 3 in...
-
On January 1, two years ago, Parkway Corporation purchased all of the outstanding common stock of Shaw Company for $220,000 cash. On that date, Shaw's net assets had a book value of $148,000....
-
Mulligan Manufacturing Company uses a job order cost system with overhead applied to products at a rate of 150 percent of direct labor cost. Required: Treating each case independently, selected from...
-
The Polaris Company uses a job-order costing system. The following transactions occurred in October: a. Raw materials purchased on account, $210,000. b. Raw materials used in production, $189,000...

Study smarter with the SolutionInn App


