Gas is bled from a tank. Neglecting heat transfer between the gas and the tank, show that
Question:
Gas is bled from a tank. Neglecting heat transfer between the gas and the tank, show that mass and energy balances produce the differential equation:
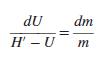
Here, U and m refer to the gas remaining in the tank; H′ is the specific enthalpy of the gas leaving the tank. Under what conditions can one assume H′ = H?
Transcribed Image Text:
dU dm H' - U m
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
The heat transfer between the gas and the tank is neglected So No heat tra...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
Under what conditions can trespass arise?
-
Under what conditions can a project be considered as both a success and a failure at the same time?
-
Under what conditions can a company net its current deferred tax assets with its current deferred tax liabilities on the balance sheet?
-
Jen and Barry's Ice Milk Company used cash to purchase a new ice milk mixer on January 1, 2013. The new mixer is estimated to have a 20,000 - hour service life. Jen and Barry's depreciates equipment...
-
1. The CEO at 3M has formulated a new strategy of diversification with increased growth and profitability. In what ways will the new strategy affect the organization structure at 3M? How will it...
-
Refer to Exhibits. Describe managements process for evaluating internal control over financial reporting. For the control environment principles, identify evidence that management might obtain to...
-
Prepare headings for a sales journal like the one in Exhibit E-A.1. Journalize the March transactions shown in Exercise E-1 that should be recorded in the sales journal assuming that the periodic...
-
Chilczuk, S.A., of Gdansk, Poland, is a major producer of classic Polish sausage. The company uses a standard cost system to help control costs. Manufacturing overhead is applied to production on the...
-
XBRL is made up of two parts: a- Tag and barcoding b- Taxonomies and instance document c- Reporting concepts and schema d- US GAAP and IFRS 15- Each XBRL taxonomy is connected to a number of ......."...
-
A boat is pulling a parasail and rider at a constant speed. Knowing that the rider weighs 550 N and that the resultant force R exerted by the parasail on the towing yoke A forms an angle of 65°...
-
We further explore the relationship between respondents education and mothers education, computing regression models separately for males and females. 1. Calculate the regression equation for each....
-
Find an equation for the work of reversible, isothermal compression of 1 mol of gas in a piston/cylinder assembly if the molar volume of the gas is given by where b and R are positive constants. RT...
-
Ennis Inc.s Forms Solutions Group (www.ennis.com/) is a Texas-based machine-intensive printing company that produces business forms. The resources demanded by a specific job depend on the type and...
-
Coronado Corporation accumulates the following data relative to jobs started and finished during the month of June 2025. Costs and Production Data Actual Standard Raw materials unit cost $2.20 $2.10...
-
Consider the following labor statistics for the adult population (age 16 and older) in Norway displayed in the table below (all numbers in millions). Employed 110 13 Not in Labor Force 75 Unemployed...
-
Blossom Variety Store uses the LIFO retail inventory method. Information relating to the computation of the inventory at December 31, 2026, follows: Cost Retail Inventory, January 1, 2026 $147,000...
-
Use polynomial division to show that the general expression for the factors of the difference of two cubes, x 3 - y 3 = ( x - y ) ( x 2 + xy + y 2 ) , is correct
-
A beam has a lenght of L = 9 m long and carrying the uniformly distributed load of w = 3 kN/m. w kN/m A RA a) Calculate the reaction at A. RA= KN L RB b) Calculate the maximum bending moment. Mmax=...
-
Abercrombie & Fitch Co. (ANF) is a specialty retailer of casual apparel. The following is a series of accounts for ANF. The accounts are listed alphabetically and numbered for identification....
-
Repeat Exercise 16.6 using the t-test of the coefficient of correlation. Is this result identical to the one you produced in Exercise 16.6?
-
Calculate the theoretical flame temperature when ethylene at 25(C is burned with: (a) The theoretical amount of air at 25(C. (b) 25% excess air at 25(C. (c) 50% excess air at 25(C. (d) 100% excess...
-
For steady Bow through a heat exchanger at approximately atmospheric pressure, what is the final temperature, (a) When heat in the amount of 800 kJ is added to 10 mol of ethylene initially at 200(C?...
-
What is the standard heat of combustion of n-pentane gas at 25(C if the combustion products are H2O(l) and CO2(g)?
-
Duncan Inc. issued 500, $1,200, 8%, 25 year bonds on January 1, 2020, at 102. Interest is payable on January 1. Duncan uses straight-line amortization for bond discounts or premiums. INSTRUCTIONS:...
-
WISE-HOLLAND CORPORATION On June 15, 2013, Marianne Wise and Dory Holland came to your office for an initial meeting. The primary purpose of the meeting was to discuss Wise-Holland Corporation's tax...
-
Stock in ABC has a beta of 0.9. The market risk premium is 8%, and T-bills are currently yielding 5%. The company's most recent dividend is $1.60 per share, and dividends are expected to grow at a 6%...

Study smarter with the SolutionInn App


